Page 1, 2, 3
Effects of Addiction of Endocrine Pathology
Effects on HPA, Pituitary, and Sex Hormones
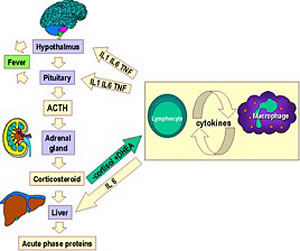
It is documented that prolonged elevation of glucocorticoid level is associated with persistent, chronic pain or stress disorders. Furthermore, in certain individuals, the administration of opioids for pain leads to negative effects on the HPA axis. Moreover, chronic opioid use and or abuse leads to decreased glucocorticoid response to acute activation of the hypothalamus-pituitary-adrenal axis, leading to adrenal insufficiency. It is worth noting that opioids exert direct stimulatory effects on adrenal glucocorticoid secretion via µ- and k-receptors.47,48
The influential role of opioids on the hypothalamic-pituitary-gonadal HPG axis was studied extensively. A dose-dependent response was observed between morphine and LH release in experimental animals.49,50
Opioids can effectively inhibit hypothalamic GnRH secretion and suppress LH levels in humans.51 Chronic morphine abuse is related to inhibition of GnRH secretion in humans.52 Li and Pelletier reported that morphine can reduce the biosynthesis of GnRH by downregulation of GnRH mRNA levels.53 Opioids can regulate biosynthesis of gonadal sex steroidal hormones, the end products of the HPA axis via the feedback inhibition process. In experimental castrated-animal studies, estradiol- or testosterone-induced LH decrease was reversed by naloxone administration.54
Chronic morphine abuse has no direct effect on serum LH levels. However, it can increase the hypothalamus sensitivity to the testosterone-related negative feedback mechanism. In females, prolonged opioid abuse leads to estradiol–surge induced LH hypersecretion and increases estradiol-mediated negative feedback mechanisms. Thus, it is clear that morphine can amplify positive and negative feedback on gonadotropin release.55,56
Opioids, including exogenous and endogenous, can influence the increase and decrease of gonadal steroid hormones. Opioid peptides can cause decrease in LH pulse frequency.57 In female experimental animals, central opioid neuron-mediated tonic inhibition of LH levels significantly affected the luteal phase. This leads to reduced LH surge and associated amenorrhea or oligomenorrhea.58 Effects on opioids on the HPG axis can reduce the LH levels in humans.59,60 The negative effects of opioids on the HPG axis depend on the circulatory levels of sex steroids during the menstrual cycle.61 The adverse events of opioids on the HPG axis vary with age and increase during the pubertal stage.62 During early and middle puberty, the negative feedback mechanism of estradiol on the HPG axis is independent of opioid receptor pathways.63 In humans, chronic opioid administration leads to reduced LH levels; however, the serum FSH levels remain normal. Studies have confirmed that opioids can affect LH release only in the presence of sex steroids. This is due to negative feedback inhibition of hypothalamus GnRH secretion.64 It is now clear that sex steroidal hormones are essential for modulation of opioid sensitivity in the HPG axis.
Effects of Opioids on Arginine-Vasopressin
Effects of Narcotics on Oxytocin
Opioids selectively inhibit oxytocin secretion in humans. Prolonged morphine abuse could affect biosynthesis and release of oxytocin. Opioids suppress oxytocin regulation by directly acting at the pituitary and hypothalamic regions with key involvement of both k- and µ-receptors.
Effects of Addictive Drugs on the Pathophysiology of Thyroid Hormones
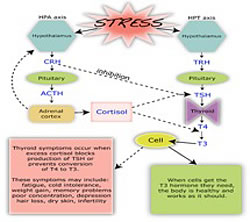
In humans, administration of morphine could significantly increase the TSH levels among normal and hypothyroid individuals.65 The TSH-stimulatory effects of morphine were confirmed by several other studies.66 When compared with normal individuals and cigarette smokers, decreased levels of TSH were observed among opium users. The underlying pathophysiological mechanism could be stress conditions.67
Opioids stimulate TSH secretion in humans due to interaction with endogenous enkephalins. The hypothalamus is the key site for opioid action on the hypothalamus-pituitary-thyroid (HPT) axis. k-receptors are the primary binding sites involved in the opioid action on TSH.
 |
 |
Roles of Addictive Substances on Sex Hormonal Disorders+
Administration of morphine reduces LH secretion as a result of decreased concentrations of sex steroidal hormones. Opioids significantly impair female menstrual cycle by suppression of progesterone, LH, estradiol and FSH levels.68 Irregular menstrual cycle with amenorrhea is a prominent symptom of hypogonadism in women.
Opioids cause decreased synthesis of adrenal androgen production and androgen-dependent loss of libido. Controlled clinical trials have suggested that prolonged use of heroin could affect sexual desire and performance in men.
In heroin- and methadone-addicted men, decreased levels of testosterone were reported. About 89% of male hypogonadism is associated with oral opioid abuse. Oral opiates can significantly reduce FSH, LH, estradiol, and dihydrotestosterone among men.69.70 With addition of hypogonadism and loss of libido, opiates can cause symptoms of depression.71,72 Depression with sexual–dysfunction related stress is a key risk for addictive behaviors that can drive the individuals towards habit-forming drugs such alcohol, morphine, marijuana, and heroin.
This review article clearly indicates how complex is the stress, pain, addiction, and neuroendocrine interplay. Therefore a more comprehensive wholesome understanding of this stress, survival, and reward symphony is a must for us to effectively address and cure this disease of pain, stress, and addiction imbalance. I therefore propose an integrative addiction educational forum led by the experts in natural addiction therapy.
Page 1, 2, 3 |
![]()
![]()
![]()







