|
Page 1, 2, 3
With the fast-moving life of today, one tends to find oneself always trying to cope with constant stressors. The fast evolution and constant change that characterizes this era of rapid technology has altered the fabric of our society. Everything and everyone has evolved abruptly, especially over the past 10 years or so. This hasty shift in technology, work conditions, family makeup, and expectations can easily create very steep imbalances, predisposing individuals to fall into what we call stress. Stress may be triggered by the slightest event, including family, friends, and peer pressure. Our society is inflamed; hence stress and pain are becoming fuel for the addiction epidemics.
The debilitating stress effects of pain have also significantly contributed to an indoctrinatory overprescription pattern of painkillers. In particular, opioid prescription and abuse have increased in the last decades. Prescription of opioids for management of noncancer chronic pain is increasing over the years, providing the main source of opioid access. The long-term effects of opioids use on the metabolism, particularly the endocrine system, is one of the prime foci of recent research studies. Opioids act on the hypothalamic-pituitary-adrenal (HPA) axis; increased levels of prolactin and GH; and decreased oxytocin, testosterone, LH, and estradiol in humans. However, the effects of opioids on arginine vasopressin and ACTH are conflicting.
Stress, Reward System, and Dopaminergic Pathway
Stress can be defined as any stimulus that alters physiological and psychological homeostasis or equilibrium. The nature and effects of stress may vary that stimulate or suppress several molecular and/or cellular signalling molecules. The stress response generally varies from one person to another due to distinctive coping styles.1,2 Acute and chronic stress may lead to physiological and psychological disorders, including predisposition to addiction.3
Humans respond to stress via the HPA axis, activation of corticotrophin-releasing factor outside the hypothalamus and activation of sympathetic nervous system via adrenaline or noradrenaline.4–6
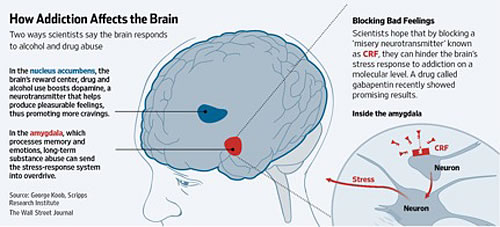
Alcohol and stress can induce the hypothalamus to release corticotrophin-releasing hormone (CRH) that facilitates release of b-endorphin. CRH is then transported to the anterior pituitary gland that synthesizes proopiomelanocortin (POMC), a key factor for stress hormones including ACTH and beta-lipotropin. ACTH is a potent stimulator of cortisol release. Other stress inducers, including noradrenaline, serotonin, and GABA, regulate CRH release. Similar pathophysiological changes are observed in addicted individuals.7–10 In stress conditions, sympathetic nerves contribute to adrenaline and noradrenaline release from the adrenal gland and cause stress.11,12
Increased CRF production outside the hypothalamus stimulates the mesocorticolimbic dopaminergic system, which is dopamine dependent. This system includes nucleus accumbens, amygdala, hippocampus, and ventral tegmental area (VTA). In addictive individuals, the brain's reward pathway is primarily mediated by the mesocorticolimbic dopamine system. Apart from addiction, the limbic system and hypothalamus are involved in the stress response and stimulation of CRF activity.13–15
Dopamine Levels in Acute Stress and Chronic Stress
Preclinical and human clinical studies have demonstrated the influence of stress on dopamine synthesis or mesocorticolimbic dopaminergic activity. Radiological studies have demonstrated that amphetamine, alcohol, and other addictive drugs have triggered dopamine release.16–18 The pathophysiological relationship between stress hormones, drug addiction, and mesocorticolimbic dopamine activity is well documented.
Pruessner and colleagues have confirmed that stress during early stages of life could promote dopamine release in the ventral striatum that causes psychosocial stress and addiction.19 The study results were confirmed by Oswald et al., suggesting the links between stress-associated cortisol release, dopamine levels, and amphetamine.20 Psychological studies have confirmed that cortisol is a key contributor for dopamine release in stressed individuals.21 However, the levels of cortisol and dopamine vary in acute and chronic stress conditions.
In adrenalectomized experimental animals, the body's stress response via the HPA axis contributes to glucocorticoids and dopamine suppression in the nucleus accumbens.22,23 When the adrenal glucocorticoids were replaced with corticosterone injections, normal dopamine levels were restored. These studies have suggested the role of adrenals on dopamine release; acute stress could moderately affect the adrenals, and contributes to moderate dopamine increase. However, in chronic stress, it could be vice versa.
In acute stress, the expression and activity of dopamine is comparatively less than in chronically stressed individuals. The negative effects in the former are generally reversible and moderate. However, in chronically stressed individuals, the altered dopamine levels lead to hormonal, psychological, and behavioral dysregulation that increases the predisposition to addiction.
Dopamine Receptor Levels in Acute and Chronic Stress
The effects of dopamine on stress conditions depend on the expression of dopaminergic receptors. Humans exhibit genetic variability in the expression of dopaminergic receptors such as D2 and/or D3 receptors, dopamine turnover in response to stress-related stimuli.
Experimental studies have suggested that the effects of acute or chronic stress on the mesolimbic system and behavioral patterns are influenced by epigenetic patterns. Acute stressors induce 3,4-dihydroxy phenylacetic accumulation in the brain with significant reduction in dopamine levels.24
In experimental animals, repeated or prolonged stress could lead to hyposensitivity of D2 presynaptic receptors. Continuous exposure to restraint stress leads to decrease in D2 receptor density with normal levels of D1 receptor density in the nucleus accumbens. The direct relationship between the dopamine receptors and stress conditions is also confirmed by electric foot-shock tests. In experimental animals, activation of D1 and D2 receptors are vital for attenuation of fear, motor suppression, and so on.
Prenatal stress could alter the response to stress and expression of dopamine agonist/antagonist receptors. In prenatal stressed experimental animals, a significant increase in D2 receptor binding in the nucleus accumbens was observed. Significant decreases in D3 receptor binding in the core nucleus accumbens and shell were also observed.25 In some individuals, chronic stress could decrease the density of D2 receptors but not the affinity.
Dopamine Uptake in Acute and Chronic Stress
Several research studies have suggested that removal of dopamine in chronic stressed animals is an adaptive mechanism of mesoprefrontal cortex. Increased D1 receptor stimulation during stress allows the posterior cortical and subcortical structures to regulate behaviors. Stress could alter the mesoprefrontal dopamine fibers and affect biochemical responsiveness of the dopamine subcortical innervations.
In experimental animals, repeated stress reduced basal locomotor activity and utilization of dopamine in the brain. The effects could be reversed by acute administration of D1 or D2 receptor antagonists.
In prolonged, mild stress conditions, dopamine uptake levels are affected, leading to altered behaviors.26–28
Moderate uptake of dopamine is important for normal behaviors, and excessive dopamine activity or impaired uptake leads to altered spatial working memory functions. This condition is common in acutely and chronically stressed individuals. However, the uptake of dopamine differs in both stress conditions.
Dopamine Metabolism in Acute and Chronic Stress
Repeated or prolonged stress has negligible effects on dopamine and serotonin metabolism. This may be due to adaptive mechanisms of the higher centers via the stress response mechanisms. However, acute stress conditions could decrease the hypothalamic epinephrine levels and return to normal within few hours.
Dopamine metabolism is affected due to hypothalamic adrenaline concentration and turnover in response to acute and chronic stress. Impaired adrenaline and noradrenalin turnover affects dopamine metabolism and causes behavioral changes, including predisposition to addiction.29,30
As with stressed individuals, decreased dopaminergic functions are reported in cocaine addicts. This could be due to reduced D2 receptor availability or expression, or impaired dopamine metabolism in cingulate gyri, frontal lobes, and orbitofrontal cortex.
Dopamine dysmetabolism could lead to loss of control with compulsive reinforcement that predispose to addictive behaviors.31
Dopamine Factors and Predisposition to Addiction
Significant neurobiological links between stress, dopamine factors, reward pathways, and risk of addiction are well documented. The reinforcing properties of habit-causing drugs are associated with the activation of the mesolimbic dopaminergic pathways such as prefrontal cortex, ventral striatum and ventral tegmental area.32,33 Apart from stress mechanisms, the dopamine pathway is also associated with reward processing, adaptation, and learning.34
The role of dopamine factors in drug reward mechanisms is reported in opioid and alcohol abusers. This is due to activation of mesolimbic dopamine systems with drug cravings and euphoria.35–38
Experimental animal studies have suggested that stress exposure with increased glucocorticoid release could increase dopamine release in the nucleus accumbens.39 Suppression of glucocorticoids could reduce extracellular levels of dopamine in resting and during response to stress and addictive substances.40
Chronic elevation of glucocorticoids inhibits dopamine synthesis and turnover in the nucleus accumbens. This mechanism suggests that alterations in the HPA and glucocorticoid levels could affect dopamine transmission. Drug abuse, stress, and increased levels of CRF and/or glucocorticoids could increase glutamic acid activity in the ventral tegmental area. This leads to enhanced activity of dopaminergic neuron.41
Human brain imaging studies have further shown that stress-induced cortisol elevation is associated with dopamine accumulation in the ventral striatum. Some evidence also reveals that amphetamine-induced increases in cortisol are associated with both dopamine binding in the ventral striatum and ratings of amphetamine-induced euphoria.42
Drug abuse and stress activate the mesolimbic pathways, which results in synaptic adaptations in the ventral tegmental area of dopamine neurons with adaptive morphological changes in the medial prefrontal cortex areas.43 The ventral striatum is a main regulator of behavioral response, stress, and mesolimbic dopamine pathways.44 Mesolimbic dopamine pathways are linked with rewarding, stress adaptation and goal-directed behaviours. These pathways are also important for emotion control, stress processing, decision making and impulse control. Altered status leads to impaired stress processing, impulse control, and predisposition to addiction.45
Opioid abuse can impair secretion of pituitary hormones and cause hypogonadism and menstrual disorders.46 The negative interaction between addiction and the hypothalamic-pituitary-gonadal (HPG) axis is well studied.
Page 1, 2, 3
|
![]()
![]()
![]()





