|
![]()
From the Townsend Letter |
|||||
Blood-Brain Barrier Damage and Neuroautoimmunity Aristo Vojdani, PhD, MSc |
|||||
|
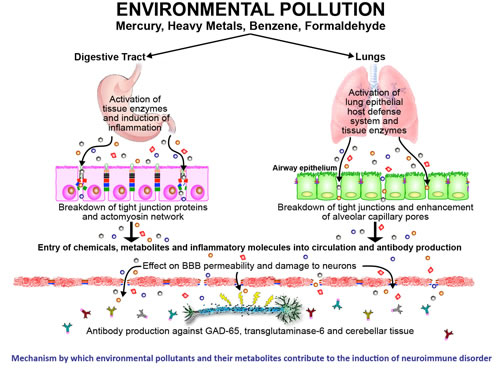
Environmental Exposures These autoantibodies can attack key nervous system components such as GAD-65, transglutaminase-6, and cerebellar tissue, potentially leading to neuroinflammatory and neurodegenerative disorders. Thus, detecting significant elevations in IgA, IgG, and IgM antibodies against barrier-forming proteins is key to understand the environmental pollutant mechanistic pathways affecting epithelial and endothelial barriers. The mechanisms by which environmental pollutants can affect the barriers and lead to neuroautoimmunity are shown in Figure 2. Figure 2. Proposed mechanism for the effect of environmental pollutants on the BBB, leading to neuroautoimmunity. Environmental pollutants enter the system either through inhalation or consumption, activating enzymes and inducing inflammation. This opens the tight junctions, allowing pollutant molecules to enter the circulation, causing autoantibody production. Autoantibodies attack neuronal cells of the CNS, leading to demyelination and neuroautoimmunity.
Chemical toxins. Simply measuring chemicals in urine or blood is not enough to indicate exposure. In 80% of patients, these chemicals are successfully excreted, but in an estimated 20% of patients, chemicals form a complex with human tissue and remain in the body, possibly for life. When these chemicals bind to human tissue (for example as deposits in fat cells), they induce antibody production against both the toxins and human tissue. Research was conducted measuring antibodies in blood samples from 400 so-called healthy subjects (200 males and 200 females of different ethnicities with a median age of 36.2 years). Researchers found that 25% of participants were producing antibodies against various chemicals, indicating chronic exposure to chemical haptens. The formation of chemical-protein adducts could be one of the mechanisms by which environmental chemicals induce autoimmune reactivity. Exposure to molds and mycotoxins or infection. A significant number of patients experience symptoms in response to molds and/or mycotoxin exposure. Various other types of infectious agents can also have these effects, including Lyme disease. Autopsy research has reported that brain tissue of Lyme patients evidenced plaque and tangles typically characteristic of patients with Alzheimer's disease. Viral hitchhiking. The BBB works efficiently to prevent brain infections. However, viruses can penetrate the barrier by attaching onto circulating cells of the immune system. Systemic LPS (lipopolysaccharides) enhance both immune cell and free virus transport across the intact BBB. In vivo and in vitro studies, for example, have found that free HIV-1 can be taken up by brain endothelial cells and cross the BBB. LPS act at the luminal surface of the brain microvascular endothelial cell monolayer, which induces abluminal secretion of cytokines and other factors that subsequently act on pericytes. The pericytes then secrete substances that enhance viral transcytosis across the BBB. Electromagnetic fields.Effects of cell phone radiation on the BBB were initially identified 20 years ago. In an animal study evaluating radio frequency radiation, Adlkofer found albumin leakage in 32% of the 184 animals evaluated. Human clinical trials have shown that exposure to radio frequency radiation results in the leakage of albumin from the brain and is also associated with symptoms of memory loss and forgetfulness. Radio frequency energy is transmitted by cell phones, tablets, and laptop computers through blue tooth and other wi-fi connections. Table 1: Cross Reactivity Between Environmental Triggers and Neurological Tissues
Intestinal Permeability The gut-brain connection has been well studied in the existing literature. Gut epithelium resembles the BBB in many respects, with perhaps 75% similarity in structural composition. Due to antigen similarity between tissue proteins in gut endothelium and in BBB proteins, "leaky gut" can result in "leaky brain." If the gut barrier is not repaired, those molecules now open the BBB, resulting in inflammation and autoimmunity in the nervous system. Dysbiosis. Dysfunctional flora or overgrowth is a well-recognized trigger of leaky gut. Certain dietary components such as gluten, dairy, and other antigens can also open the BBB, resulting in antibody production against nervous system antigens and brain receptor cells. Due to the similarity between gluten and casein (both a-casein and b-casein), and synuclein and oligodendrocytes in the brain, resulting antibodies can attack nervous system antigens, resulting in neuroautoimmunity. Food antigens. LPS is not the only molecule that has been shown to transport viruses across the BBB. In an animal study, wheat germ agglutinin (WGA), a lectin protein from wheat, was injected into subjects. WGA was found to bind to sialic acid and N-acetylglucosamine, inducing vesicle-mediated internalization of WGA by brain endothelial cells, a process called adsorptive endocytosis. Lectin-induced vesicles provide a mechanism by which enveloped viruses can be internalized by cells. These study results strongly suggest that glycoprotein gp120 or gp120/gp41 induces adsorptive endocytosis and the uptake of HIV-1 by brain endothelial cells. This action explains how free, blood-borne viruses can infect the CNS while the BBB remains intact. Environmental factors, if they are powerful enough, can initiate the cascade of events that directly or indirectly opens the tight junctions. If we do not repair the gut barrier, those molecules can open the blood-brain barrier, triggering adverse effects that include inflammation, oxidative stress, neurotoxicity, autoimmunity, degenerative disease, or cancer. Advanced Antibody Testing: Array 20 Evaluating preclinical symptomology. The test has efficacy in the assessment of preclinical conditions to evaluate potential breaching of the blood-brain barrier due to trauma, environmental triggers, or dementia with useful findings even in the absence of a clearly defined diagnosis. Assessment of minor injuries. This lab work is useful for patients who play contact sports and/or have experienced trauma to determine whether a concussion or TBI has occurred. The test is cost-effective, so it can be used in situations involving minor injury such as high school athletics, for example, when an MRI would seem excessive and could be prohibitively expensive. Rehabilitation. Array 20 can also be used to monitor injuries, indicating when it will be safe to return to normal activities. One of the major concerns after a first injury is the prevention of recurrent injuries before the brain is completely healed. Repetitive brain injury that occurs before complete healing and repair of the neuroinflammatory response can accelerate and exacerbate chronic activation of the microglia, with serious consequences. Management of complex neurological disease. Antibody testing provides a noninvasive tool for assessing the effectiveness of interventions for neurodegenerative disorders such as Alzheimer's disease, amyotrophic lateral sclerosis (Lou Gehrig's disease), epilepsy, multiple sclerosis, Parkinson's disease, stroke, and vascular dementia.
Clinical Strategies Rationale for Testing Antibody testing is an evaluation technique that has demonstrated 99% sensitivity in the clinical setting. This is a viable adjunct or alternative to imaging for the full range of neuroautoimmune disorders, including traumatic brain injury, progressive dementia, neurodegenerative disease, and autoimmunity. |
|||||
![]()
Consult your doctor before using any of the treatments found within this site.
![]()
Subscriptions
are available for
Townsend Letter, the Examiner of Alternative Medicine
magazine, which is
published 10 times each year. Search our pre-2001
archives for further information. Older issues of the printed magazine
are also indexed for your convenience.
1983-2001
indices ; recent indices. Once you find the magazines you'd like to order, please
use our
convenient form, e-mail subscriptions@townsendletter.com,
or call 360.385.6021.
360.385.6021
Fax: 360.385.0699
info@townsendletter.com
Who are
we? | New articles | Featured
topics | e-Edition |
Tables of contents | Subscriptions | Contact
us | Links | Classifieds | Advertise |
Alternative
Medicine Conference Calendar | Search site | Archives |
EDTA Chelation Therapy | Home
© 1983-2014 Townsend Letter
All rights reserved.
Website by Sandy
Hershelman Designs