|
![]()
From the Townsend Letter |
||||
Blood-Brain Barrier Damage and Neuroautoimmunity by Aristo Vojdani, PhD, MSc |
||||
When the BBB is damaged, circulating antibodies that cross-react with neurological tissues can infiltrate the brain and nervous system, with the potential destruction of neurologic tissues. The cycle of neuroautoimmunity begins with a breach of the gut and/or blood-brain barriers. BBB permeability and the effects of "leaky brain" contribute to many devastating neurological and autoimmune conditions, including traumatic brain injury (TBI). New breakthroughs in laboratory testing make it possible to identify BBB permeability in trauma ranging from suspected concussions to TBI. The testing is also relevant to neurological disorders as advanced as Alzheimer's dementia and as subtle as chronic headaches or memory loss. Testing for key antibodies makes it possible to diagnose many of these conditions at a preclinical stage, before frank symptoms of injury or disease are present. Increased permeability can manifest as CNS symptoms of neurotoxicity, autoimmunity, or even cancer. Indications
Barrier Dysfunction and Disease Absence of Immune Tolerance Numerous exposures can trigger BBB permeability:
Breaches of the BBB can result in the damaging effects of TH 1 and TH 17 lymphocytes, as well as antibodies that can target and damage neurons and tissues such as myelin basic protein. Autoantibodies can become pathogenic on penetration into the CNS through either BBBD (blood-brain barrier dysfunction) or extravasation into cerebrospinal fluid from the sub-arachnoid space. Testing for Barrier Dysfunction BBB protein antibodies.The blood-brain barrier includes endothelial cells, tight junctions, the capillary basement membrane (BM), pericytes (PCs), and astrocyte end-feet that tightly ensheath the vessel walls. Astrocytes located beneath the endothelial cells prevent the entry of unwanted molecules and produce a protein identified as BBB protein, which is now used in laboratory testing as a marker to identify the presence of neuroautoimmune reactivity. Elevated antibody titers for BBB protein and other brain proteins have been described in Alzheimer's disease and epilepsy. In senile dementia, the presence of BBB protein antibodies is one of the early markers of cognitive decline. (Specific testing for BBB protein antibody is provided in Cyrex Laboratories' Array 20.) Occludin and zonulin antibodies.Paracellular space between the endothelial cells of the BBB is protected by an extensive network of tight junctions, consisting of transmembrane proteins that include occludin, claudin, and junction adhesion molecule (JAM), as well as cytoplasmic proteins, specifically zonulin-1 (ZO1), zonulin-2 (ZO2), and zonulin-3 (ZO3). Many patients with autoimmune disorders produce antibodies against their own occludin and zonulin, which serve as useful laboratory markers in diagnostic testing. (This evaluation is provided in Cyrex Array 2.) Lipophilic toxins.Tight junctions between the cells of the BBB endothelium form a physical barrier, significantly reducing passive diffusion through the paracellular pathway, and forcing any molecular traffic to occur mainly across the endothelial transcellular pathway. However, gases such as oxygen and carbon dioxide can easily diffuse across the barrier via the lipid membranes. This provides a security breach through which toxic lipophilic chemicals including petroleum-based products and other fat-soluble chemicals can cross the BBB, invading the CNS. (Antibodies targeting lipophilic toxins are among the 12 substances evaluated in Cyrex Array 11.) A growing body of research has implicated the effects of BBB dysregulation in acceleration of chronic neurodegenerative disorders, including:
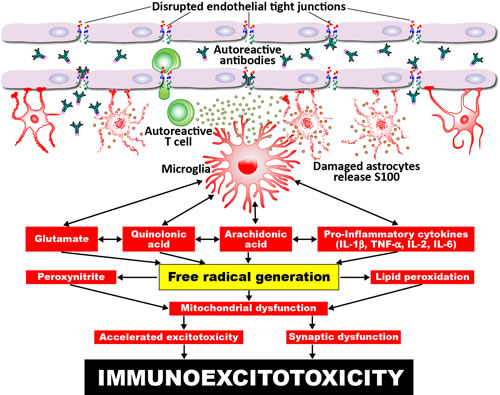
The mechanisms by which toxic chemicals disrupt the endothelial tight junctions are shown in Figure 1, through processes that result in damage to astrocytes, microglia activation, the production of autoreactive antibodies, free radical generation, and immunoexcitotoxicity. Figure 1. Proposed mechanism for excitotoxicity from leaky BBB. Environmental triggers, brain inflammation, and disruption of the BBB leads to the release of free radicals and immunoinflammatory factors, causing acute microglia activation, which contributes to immunoexcitotoxicity.
Cancer. Polychlorinated biphenyls (PCBs) are a prime example of a toxin that can disrupt blood-brain barrier integrity and promote brain metastasis formation. When these exposures occur, the chemical opens the barrier, damaging the astrocytes, which then can no longer protect the barrier. The same process can occur with infections such as Borrelia burgdorferi, cytomegalovirus, Epstein-Barr virus, herpes type 6, and other environmental triggers which directly or indirectly damage the barriers, resulting in neuroautoimmunity and even cancer. Specific Exposures
These processes have been confirmed in clinical research such as a study of 57 football players, medically evaluated before and after games, measuring the level of S100-B proteins. Serum S100-B was detected in players who experienced the greatest number of sub-concussive hits. Since this is a large protein molecule (21 kDa), even trace amounts handled by cells involved in the immune system resulted in antibodies against S100-B. The presence of this protein in a blood sample means that the BBB has been breached or damaged, and antibodies produced against S100-B have invaded the CNS. The high degree of sensitivity of this testing was confirmed in a French study involving 2,000 patients with minor head injuries, comparing CT scans with plasma S100-B levels. TBIs were identified by antibody testing with 99% sensitivity and 20% specificity, confirmed by CT scan with the conclusion that S100-B testing is a highly promising screening tool.
|
||||
![]()
Consult your doctor before using any of the treatments found within this site.
![]()
Subscriptions
are available for
Townsend Letter, the Examiner of Alternative Medicine
magazine, which is
published 10 times each year. Search our pre-2001
archives for further information. Older issues of the printed magazine
are also indexed for your convenience.
1983-2001
indices ; recent indices. Once you find the magazines you'd like to order, please
use our
convenient form, e-mail subscriptions@townsendletter.com,
or call 360.385.6021.
360.385.6021
Fax: 360.385.0699
info@townsendletter.com
Who are
we? | New articles | Featured
topics | e-Edition |
Tables of contents | Subscriptions | Contact
us | Links | Classifieds | Advertise |
Alternative
Medicine Conference Calendar | Search site | Archives |
EDTA Chelation Therapy | Home
© 1983-2014 Townsend Letter
All rights reserved.
Website by Sandy
Hershelman Designs