Page 1, 2
Urine Testing and Oral Hormone Delivery
Like serum, urine hormone test results can be misleading when hormones are delivered orally. As mentioned above, when a hormone is introduced into the gastrointestinal tract, much of it is metabolized to downstream phase I and II metabolites/conjugates, which are rapidly eliminated in urine.11,15,17 Urine testing measures the total amount of metabolites that enter urine, which is more reflective of the amount of hormone dosed and not the actual amount of bioactive hormone that enters tissues.
Since it takes about 10× physiological dosing of any orally delivered hormone (e.g., about 100–300 mg of oral progesterone) to get enough of the bioactive hormone into the systemic circulation for a biological effect, this results in very high circulating levels of hormone metabolites, which have no biological effect and are rapidly removed from circulation and excreted into urine.10,11,15,17
Figure 2 (determined at ZRT Laboratory using GC/MS/MS) shows the median ranges for urinary pregnanediol, a progesterone metabolite commonly used to estimate, indirectly, progesterone levels in urine in premenopausal and postmenopausal women and those supplementing with different forms of progesterone.10,17 Urinary pregnanediol levels, as expected, are low in premenopausal women during the follicular phase of the menstrual cycle, but increase significantly during the luteal phase, as reported by others.17 Also as expected, pregnanediol levels are low in postmenopausal women and women using contraceptive synthetic progestins. Women using 10 to 50 mg of topical progesterone or 100 to 300 mg of vaginal progesterone suppositories have little pregnanediol in urine, which is why we do not recommend using urine to monitor any form of topically delivered hormone. Progesterone levels in urine following topical delivery are also low, and urinary progesterone levels with vaginal progesterone fluctuate erratically due to direct contamination of the urine (not shown). Pregnanediol remains low with vaginal delivery, but this is problematic because it does not accurately reflect the tissue distribution of progesterone seen in saliva and DBS assays.
Figure 2
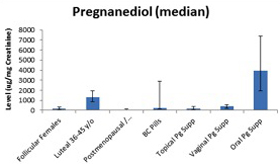
Levels of urinary pregnanediol, following 100 to 300 mg oral dosing, are much higher than levels seen in women during the luteal phase of the menstrual cycle, which taken at face value could be misconstrued as overdosing. This isn't likely for several reasons: first, levels of serum, saliva, and bloodspot are not elevated with oral progesterone dosing; second, clinically, this is an appropriate dose.
Urine testing of the supplemented hormone therefore reveals approximately how much hormone was consumed and eliminated but does not provide insight into how much of the active hormone was present in the systemic circulation or entered target tissues. Therefore, while urine testing is an excellent way to evaluate endogenous production of the sex hormones (Figure 1), it will not likely be useful clinically as a diagnostic fluid for exogenous oral or topical hormone delivery.
Conclusions
In summary, each of the four body fluids can be used as a diagnostic medium when hormones are produced endogenously. With oral administration of the sex hormones, accurate testing of hormones in serum requires extraction and separation of metabolites from the parent steroid. Testing in urine is not recommended unless the range is reestablished for the common dosing (e.g., 100–300 mg oral progesterone) and the range readjusted upwards to reflect the expected level. Serum/plasma and urine are not recommended body fluids for testing steroid hormones delivered by topical route of administration.
Page 1, 2
Notes
1. Zava DT. The trouble with topical progesterone and testing. Townsend Lett. 2014 January.
2. Du JY, Sanchez P, Kim L, Azen CG, Zava DT, Stanczyk FZ. Percutaneous progesterone delivery via cream or gel application in postmenopausal women: a randomized cross-over study of progesterone levels in serum, whole blood, saliva, and capillary blood. Menopause. 2013;20:1169–1175.
3. Stanczyk FZ, Cho MM, Endres DB, Morrison JL, Patel S, Paulson RJ. Limitations of direct estradiol and testosterone immunoassay kits. Steroids. 2003;68:1173–1178.
4. Yang DT, Owen WE, Ramsay CS, Xie H, Roberts WL. Performance characteristics of eight estradiol immunoassays. Am J Clin Pathol. 2004;122:332–337.
5. Wang C, Catlin DH, Demers LM, Starcevic B, Swerdloff RS. Measurement of total serum testosterone in adult men: comparison of current laboratory methods versus liquid chromatography-tandem mass spectrometry. J Clin Endocrinol Metab. 2004;89:534–543.
6. Edelman A, Stouffer R, Zava DT, Jensen JT. A comparison of blood spot vs. plasma analysis of gonadotropin and ovarian steroid hormone levels in reproductive-age women. Fertil Steril. 2007;88:1404–1407.
7. Glaser R L, Zava DT, Wurtzbacher D. Pilot study: absorption and efficacy of multiple hormones delivered in a single cream applied to the mucous membranes of the labia and vagina. Gynecol Obstet Invest. 2008;66:111–118.
8. Shirtcliff EA, Reavis R, Overman WH, Granger DA. Measurement of gonadal hormones in dried blood spots versus serum: verification of menstrual cycle phase. Horm Behav. 2001;39:258–266; Petsos P, Ratcliffe WA, Heath DF, Anderson DC. Comparison of blood spot, salivary and serum progesterone assays in the normal menstrual cycle. Clin Endocrinol (Oxf). 1986;24:31–38.
9. Metcalf MG, Evans JJ, Mackenzie JA. Indices of ovulation: comparison of plasma and salivary levels of progesterone with urinary pregnanediol. J Endocrinol. 1984;100:75–80.
10. Levine H, Watson N. Comparison of the pharmacokinetics of crinone 8% administered vaginally versus prometrium administered orally in postmenopausal women. Fertil Steril. 2000;73:516–520.
11. Kushnir MM, Rockwood AL, Bergquist J, et al. High-sensitivity tandem mass spectrometry assay for serum estrone and estradiol. Am J Clin Pathol. 2008;129:530–539.
12. Simon JA. Micronized progesterone: vaginal and oral uses. Clin Obstet Gynecol. 1995;38:902–914.
13. Hargrove JT, Maxson WS, Wentz AC, Burnett LS. Menopausal hormone replacement therapy with continuous daily oral micronized estradiol and progesterone. Obstet Gynecol. 1989;73:606–129.
14. Nahoul K, Dehennin L, Jondet M, Roger M. Profiles of plasma estrogens, progesterone and their metabolites after oral or vaginal administration of estradiol or progesterone. Maturitas. 1993;16:185–202.
15. Vining RF, McGinley RA. The measurement of hormones in saliva: possibilities and pitfalls. J Steroid Biochem. 1987;27:81–94.
16. Stanczyk FZ, Gentzschein E, Ary BA, Kojima T, Ziogas A, Lobo RA. Urinary progesterone and pregnanediol. Use for monitoring progesterone treatment. J Reprod Med. 1997;42:216–222.
 Dr. David Zava has devoted his 40-year professional career to exploring the role of hormones in aging and disease. After completing his PhD in biochemistry at the University of Tennessee in 1974, Dr. Zava spent much of his time researching hormones and breast cancer in Switzerland, Texas, California, and Oregon. In 1998, he established ZRT Laboratory, a CLIA-certified laboratory that is a front-runner in the innovative development of test methods to identify hormonal imbalances that can lead to debilitating symptoms, diminished quality of life, and increased risk for cancers and many of the diseases of aging, such as diabetes, cardiovascular disease, and senile dementia. ZRT Laboratory was one of the first laboratories to develop and commercialize noninvasive saliva and dried urine and semi-invasive dried blood spot methods for testing hormones as an alternative to conventional serum testing. Using these unique methods of body fluid collection, ZRT is actively engaged in hormone research studies with universities, government agencies such as the NIH and CDC, military agencies, private physicians, and professional sports teams. Dr. David Zava has devoted his 40-year professional career to exploring the role of hormones in aging and disease. After completing his PhD in biochemistry at the University of Tennessee in 1974, Dr. Zava spent much of his time researching hormones and breast cancer in Switzerland, Texas, California, and Oregon. In 1998, he established ZRT Laboratory, a CLIA-certified laboratory that is a front-runner in the innovative development of test methods to identify hormonal imbalances that can lead to debilitating symptoms, diminished quality of life, and increased risk for cancers and many of the diseases of aging, such as diabetes, cardiovascular disease, and senile dementia. ZRT Laboratory was one of the first laboratories to develop and commercialize noninvasive saliva and dried urine and semi-invasive dried blood spot methods for testing hormones as an alternative to conventional serum testing. Using these unique methods of body fluid collection, ZRT is actively engaged in hormone research studies with universities, government agencies such as the NIH and CDC, military agencies, private physicians, and professional sports teams.
In addition to his innovations in clinical laboratory testing and development, and numerous scientific publications, Dr. Zava coauthored a landmark book, What Your Doctor May Not Tell You About Breast Cancer: How Hormone Balance Can Help Save Your Life. In this book, Dr. Zava and coauthors describe how breast cancer can be caused by hormonal imbalances that occur naturally as we age, but can be prevented if the types of hormonal imbalances are identified with testing and restored to optimal healthy levels with bioidentical hormone replacement therapy and improved lifestyle.
|
![]()
![]()
![]()






 Dr. David Zava has devoted his 40-year professional career to exploring the role of hormones in aging and disease. After completing his PhD in biochemistry at the University of Tennessee in 1974, Dr. Zava spent much of his time researching hormones and breast cancer in Switzerland, Texas, California, and Oregon. In 1998, he established ZRT Laboratory, a CLIA-certified laboratory that is a front-runner in the innovative development of test methods to identify hormonal imbalances that can lead to debilitating symptoms, diminished quality of life, and increased risk for cancers and many of the diseases of aging, such as diabetes, cardiovascular disease, and senile dementia. ZRT Laboratory was one of the first laboratories to develop and commercialize noninvasive saliva and dried urine and semi-invasive dried blood spot methods for testing hormones as an alternative to conventional serum testing. Using these unique methods of body fluid collection, ZRT is actively engaged in hormone research studies with universities, government agencies such as the NIH and CDC, military agencies, private physicians, and professional sports teams.
Dr. David Zava has devoted his 40-year professional career to exploring the role of hormones in aging and disease. After completing his PhD in biochemistry at the University of Tennessee in 1974, Dr. Zava spent much of his time researching hormones and breast cancer in Switzerland, Texas, California, and Oregon. In 1998, he established ZRT Laboratory, a CLIA-certified laboratory that is a front-runner in the innovative development of test methods to identify hormonal imbalances that can lead to debilitating symptoms, diminished quality of life, and increased risk for cancers and many of the diseases of aging, such as diabetes, cardiovascular disease, and senile dementia. ZRT Laboratory was one of the first laboratories to develop and commercialize noninvasive saliva and dried urine and semi-invasive dried blood spot methods for testing hormones as an alternative to conventional serum testing. Using these unique methods of body fluid collection, ZRT is actively engaged in hormone research studies with universities, government agencies such as the NIH and CDC, military agencies, private physicians, and professional sports teams.