Page 1, 2, 3
Protocol, Materials, and Dose Escalation
The principle of the MAP treatment is to give the two essential nutrient precursors for the endogenous production of ACh. These two nutrients are choline and pantothenic acid or pantethine. At various times we have preferred various forms of these two essential ingredients.
Our current preference is to implement the protocol using choline bitartrate and pantethine.
Our preference for choline bitartrate developed due to its high solubility in water, its potency, and the challenges in sourcing a lecithin product that is not genetically modified. Furthermore, a significant number of patients are soy or egg sensitive and most lecithin products in the marketplace are soy or egg derived. Our preference for pantethine over pantothenic acid (PA) comes from our observation that some patients do not respond to PA, though they do respond to pantethine.
We used to begin the protocol with PA due to the fact that it is considerably less expensive than pantethine; if a patient did not respond to PA, we would then switch to pantethine. We now tend to work in the reverse. We know that if we escalate through the dosing stages of MAP using pantethine and the arrhythmia does not come under control, MAP will not likely work for that patient. Conversely, if we can gain control of the arrhythmia using pantethine and choline, we can then try a switch to PA if the patient desires a lower cost option.
Achieving a delivery system which keeps the substrates at constant blood levels is a challenge. As discussed, if we want to ensure that adequate levels of precursors are available to be therapeutic, they must persist at reasonable blood concentrations at all times. We have found only one way to provide this type of steady state and that is to dose the nutrients in a beverage which the patient can drink continuously throughout the day.
We instruct patients to mix their nutrients in a 32 fluid ounce (fl. oz.), drinking bottle with water at the beginning of the day. Assuming that the patient is awake for 16 hours per day, we divide the day into 4-hour segments. During the first 4 hours of the day the patient drinks 4 fl. oz. of the beverage, between hours 5 to 8 they drink 8 fl. oz., during hours 8 to 12 they drink another 8 fl. oz., and during their final 4 hours awake they drink 12 fl. oz., ensuring adequate levels overnight (Table 1).
The MAP dosing works in a three-stage dose escalation (Table 1). At stage 1 we deliver 500 mg of pantethine and 2000 mg of choline per day. Due to the very short half-life of ACh we are able to tell within one week whether this dose is adequate. If the patient achieves complete symptomatic or objective improvement over the course of the first week then the MAP is maintained at stage 1; if not, we escalate to stage 2. Stage 2 dosing is 1000 mg of pantethine and 4000 mg of choline; after one week we reassess. If resolution of symptoms or objective findings is complete or near complete we stop at stage 2, otherwise we escalate to stage 3. Stage 3 dosing is 1500 mg of pantethine and 6000 mg of choline. Reassessment is made after one week in the same manner as previous stages. It is rare that more than 1500 mg of pantethine will demonstrate further benefit, though we have had patients who needed to escalate the initial dose of choline up to 10 grams before coming under rhythm control. After several weeks of a persistent symptom-free/arrhythmia-free period, a down titration can be attempted to find the lowest effective dose that maintains the arrhythmia resolution. When PA is substituted for pantethine, it is used at double the dose of pantethine used for that stage: 1000 mg at stage 1, 2000 mg at stage 2, 3000 mg at stage 3. When lecithin is substituted for choline bitartrate we use a non-GMO soy lecithin which contains 1000 mg of choline per tablespoon (Table 1).
Table 1: MAP Dosage Forms, Dose Escalation, and Delivery Method
Pantothenic acid/Pantethine Lecithin/Choline Bitartrate
Stage 1 1000 mg/500 mg 2 Tablespoons/2000 mg
Stage 2 2000 mg/1000 mg 4 Tablespoons/4000 mg
Stage 3 3000 mg/1500 mg 6 Tablespoons/6000 mg
*** Mixed with a sufficient quantity of water to make 32 fluid ounces (fl. oz.).
This beverage is drunk throughout the day according to the follow schedule:
Awake Hours Drink this Amount
Hours 1–4 4 fl. oz.
Hours 5–8 8 fl. oz.
Hours 9–12 8 fl. oz.
Hours 13–16 12 fl. oz.
Indications
MAP is indicated for any tachyarrhythmia, including sinus tachycardia, atrial tachycardia, supraventricular tachycardia, atrial fibrillation, atrial flutter, and accelerated ventricular rhythms, provided there are no contraindications with other aspects of treatment. MAP is also indicated for ectopic arrhythmias such as premature atrial contractions (PACs) and premature ventricular contractions (PVCs). MAP is appropriate as adjuvant therapy in the vast majority of circumstances; however, potential interactions with other treatments and medications should always be scrutinized. Patients should be closely monitored for adverse events during initiation and dose escalation. Several noncardiac indications for MAP also exist, including anxiety, hyperactivity, reactive attachment disorder, PTSD, and other deficits of neurotransmitter and ANS balance.
Side Benefits
There are several potential side benefits to supplementation with choline and pantethine or PA. Patients who take methotrexate or who have defects in folate or choline metabolism are known to have a higher dietary requirement for choline.7 Those who take oral contraception, estrogen, or progestin may have an increased requirement for pantothenic acid.5
Choline is important in several aspects of cardiovascular health. It is vital in the endothelial functions that regulate blood pressure; choline insufficiency has been implicated in atherosclerosis, and it has been shown to lower levels of homocysteine.8,30–32 It may help to control severity and need for medication in asthma.33,34 Choline may lower the incidence of neural tube defects.14,35 It may have a positive effect on several neurological conditions and processes, including psychiatric disorders, dementia, memory, and cognition.36–39 Diets deficient in choline induce fatty liver disease, and oral consumption of choline has been shown to help reverse fatty liver disease in several studies.40–42 Therefore, it is an important therapy in patients with this condition or at risk for it due to other conditions such as metabolic syndrome. Several studies have linked the importance of dietary choline to the prevention of colon, breast, and other cancers.43–45 Pantethine may improve responses to stress.46 Pantethine has been shown to improve LDL cholesterol and triglyceride levels in repeated research at doses of at least 600 to 900 mg/day.47–51
Contraindications and Side Effects
It is theoretically contraindicated to use MAP in cases of bradycardia and bradydysrhythmias or in conditions in which increasing the refractory period of the SA or AV node would be contraindicated such as first, second, or third degree AV heart blocks, SA node exit blocks, sinus pauses, and sinus arrest. There are no known interactions between choline and other supplements, drugs, diseases, conditions, or lab tests.52 The most concerning possibility for interactions between pantethine or PA and other drugs, supplements, and conditions is in anticoagulation/antiplatelet therapy and bleeding disorders.53,54 Extreme caution should be used in these circumstances and best practices would dictate counseling patients to discontinue use of pantethine two weeks before surgery.53
Choline can cause adverse reactions including sweating, gastrointestinal distress, nausea, vomiting, diarrhea, and a fishy body odor.7 Guidelines indicate that daily choline doses should not exceed 3500 mg per day in adults; however, doses of 12,000 to 16,000 mg/day have been used in a study on seizure disorders and doses of 7500 mg/day in a study on high blood pressure, both without significant adverse events.7,55 A single prospective investigation found an association between high choline diets and colorectal adenomas in women, though this observation may have been attributable to any of several other factors.56 People with liver or kidney disease, Parkinson's disease, depression, or trimethylaminuria may be at an increased risk of adverse events when consuming choline at a level up to or near the upper limit, though otherwise there is no known toxicity of choline to humans.7,57 It is regarded as likely to be safe in pregnancy and lactation when the upper limit is not exceeded.52
Pantethine is exceptionally well tolerated at doses up to 1200 mg.5,47–51,53,54 The most common side effects are gastrointestinal in nature and include nausea, diarrhea, and epigastric discomfort. When using doses in excess of 1200 mg, monitor closely and down titrate or discontinue when necessary for side effect control. There is insufficient evidence to support the safety of its use in pregnancy and lactation and should therefore be avoided.53 There is no known toxicity to humans even at very high levels of intake.5,53
Patient Data
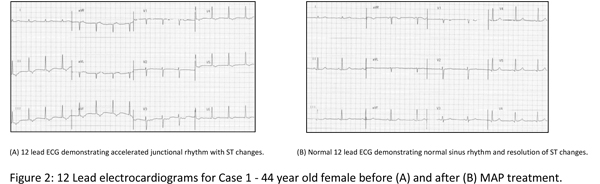
We have used the MAP protocol to address tachyarrhythmias, including atrial fibrillation, ectopic rhythms, and palpitations. In the upcoming months, we will publish a peer-reviewed case series on the success of the use of MAP in patients with these conditions. A small sampling of successful case data will be discussed here and is summarized in Table 2.
Case 1
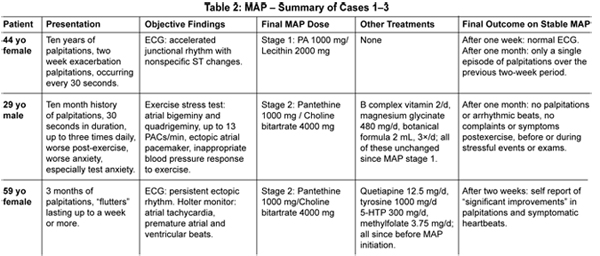
C. F., a 44-year-old female, presented to clinic with complaints of palpitations, pounding heart, and rapid heart rate. Occasional "flip-flop" sensations in her heart had occurred for more than 10 years, but had increased in frequency to every 30 seconds over the previous two weeks. Her physical exam was normal across seven systems. Her electrocardiogram (ECG) revealed an accelerated junctional rhythm, diffuse nonspecific ST segment changes, and no arrhythmic beats (Figure 2A). Blood chemistries and thyroid lab tests were within normal limits. MAP was initiated at 1000 mg of pantothenic acid and two tablespoons soy lecithin mixed in water and used daily.
One week later, the patient reported noticing a reduction in heart rate within three minutes of her initial use of the MAP. She was still having occasional palpitations but much less frequently and lasting only 10 seconds; she had not been using the MAP daily. Her ECG on that date was normal, demonstrating a sinus rhythm at a rate of 60 bpm and no ST changes (Figure 2B). The patient was reminded to use the MAP on a daily basis and was maintained at stage 1.
One month after the initial visit, the patient reported only one episode of palpitations over the previous two week period which lasted less than one minute in duration. Overall, she was pleased and thought that the treatment plan had been effective.
Case 2
X. C., a 29-year-old male, presented with complaints of palpitations lasting approximately 30 seconds and occurring every other day up to 2 to 3 times daily for the previous 10 months. They felt like early, strong, or pounding beats in his chest and neck that gave him a "dropping" feeling in his chest. An exercise stress test revealed premature atrial contractions (PACs) in patterns of bigeminy and quadrigeminy,13 PACs per minute, an ectopic atrial pacemaker, and inappropriate blood pressure response to exercise.
The patient reported daily use of a B vitamin complex and 1 gram of essential fatty acids. The MAP stage 1 was initiated using pantethine 500 mg and choline bitartrate 2000 mg along with B complex twice daily, a botanical formula (two parts each Passiflora incarnata and Crataegus oxycantha, one part each of Valeriana officinalis and Leonorus cardiaca), 2 mL three times/day, and magnesium glycinate 480 mg/day.
One week later, he reported noticing improvements after the first three days of treatment. He was feeling fewer palpitations and less anxiety in stressful situations, but was still having 1 or 2 symptomatic palpitations after daily exercise. The MAP was increased to stage 2 and no other changes were made to the treatment plan.
At two-week follow up, the patient reported that his palpitations had improved further, but were not yet completely gone. He still noticed palpitations if he did not get enough rest. Their quality and severity had decreased; he now reported them as "very subtle." Stage 2 MAP was maintained.
At follow-up two weeks later, the patient reported that he had not been having palpitations. Cardiac monitoring for heart rate variability revealed no arrhythmic beats.
Case 3
G. S., a 59-year-old female, presented with "flutters" after receiving "a lot of epinephrine" during a dental procedure 3 months prior. Immediately after the procedure she began experiencing "strong and weak flutters" that could last a week or more. The severity of her palpitations had decreased somewhat over the three months, but they persisted and were worse at night. She had an irregularly irregular pulse in a recent past visit with her PCP. She presented to us for a resting ECG which demonstrated persistent nonsinus rhythm, nonspecific ST changes, no arrhythmic beats. Follow-up 24-hour Holter monitoring revealed PACs, PVCs, and short periods of atrial tachycardia. Her history was significant for PTSD treated with quetiapine 12.5 mg, L-tyrosine 1000 mg, 5-HTP 300 mg, and methylfolate 3.75 mg, daily. MAP was initiated at stage 2 using 1000 mg pantethine and 4000 mg choline bitartrate.
Two weeks later, at the first follow-up visit, the patient reported "significant improvements" in her palpitations and her experience of discernible heartbeats.
Page 1, 2, 3 |
![]()
![]()
![]()






