|
Page 1, 2
According to the Centers for Disease Control, there are approximately 300,000 cases of Lyme disease documented, with 30,000 new cases reported each year. Those statistics are likely conservative, as many physicians have not tested for Lyme in the past, and testing results have not been totally reliable. Lyme borreliosis has a worldwide distribution and is the most common vector-borne disease is the US, according to the CDC. The CDC has now upgraded Lyme disease to official epidemic status in the US.
Lyme is caused by the Borrelia burgdorferi spirochete. The spirochete's life cycle begins when larval-stage ticks are infected via the bite of an infected Ixodes tick. These larvae then mature into nymphs, which are about the size of the a pinhead. These ticks feed upon small mammals such as white-footed field mice, catbirds, squirrels, and opossums. Whereas infection in these natural hosts does not lead to disease, infection of humans can result in Lyme disease.
In January 2016, it was reported by Harvard University that Lyme–disease carrying ticks are now in half of all US counties.1 There are more than 20 species worldwide of Borrelia, with 7 identified species described in the US. The species so far identified in the US are B. burgdorferi (Bb), B. americana, B. andersonii, B. bissettii, B. californiensis, B. kurtenbachii, and most recently B. mayonii in the Midwest.2 Of these, at least 5 have been associated with Lyme disease in the US: Bb sensu stricto, B. americana, B. andersonii, B. bissettii, and B. mayonii. The additional frustrations regarding proper diagnosis and coinfections arise, as very few ticks are carrying only one infectious vector. In addition to transmitting Bb into various animal hosts, the well-known black-legged tick (Ixodes scapularis) can also transmit strains of other infections such as Anaplasma, Bartonella, Ehrlichia, and Babesia. For example, Ixodes scapularis not only can carry the Lyme spirochete, but may also be carrying anaplasmosis, babesiosis and Powassan disease.3 This makes diagnosis and identification much more difficult. According to the US Department of Health and Human Services, the black-legged tick, lone star tick, brown dog tick, groundhog tick, Gulf Coast tick, Rocky Mountain wood tick, soft tick, western black-legged tick, and American dog tick can carry tick-borne infections.
Dr. Richard Horowitz stopped calling this phenomenon Lyme disease and named it multiple systemic infectious disease syndrome (MSIDS).4 This seems to fit the diagnosis much better, as very few chronic Lyme patients only have Lyme disease. These infections attack the immune system, stimulate an inflammatory cascade, and can attack multiple systems in the body, including the skin, brain, nervous tissue, and heart.
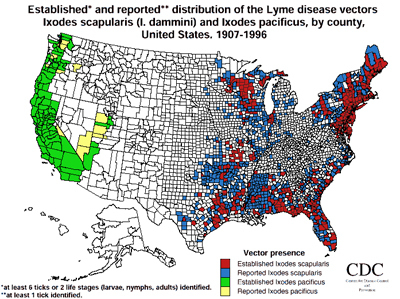
US Map of Lyme via CDC
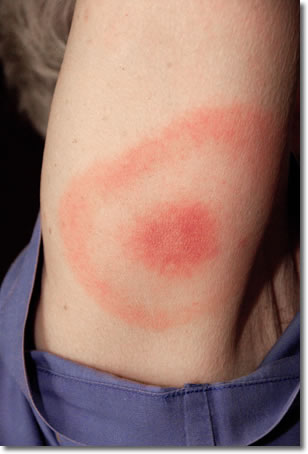 |
Erythema Migrans |
Lyme, the Infection
In 1988, Asbrink and Hovmark conceived of untreated Lyme disease as having three phases. Phase 1 represented early, localized infection, the hallmark of which was erythema migrans (EM) and its variations. This phase included fever, muscle aches, headache, nausea and fatigue. Phase 2 witnessed early dissemination of the infection with worsening of malaise and fatigue, as well as the appearance of new cardiac and arthritic signs and symptoms including AV heart block, myocarditis, migratory joint pains, and synovitis. Phase 3 saw late dissemination, with the appearance of polyneuropathy, neurocognitive and neuropsychiatric abnormalities, and meningitis, as well as chronic arthritis and debilitating fatigue.5
Lyme and the Inflammatory Pathways
Inflammation is the body's attempt at self-protection. Its goal is to remove harmful stimuli, including damaged cells, irritants, or pathogens so that the body can begin the healing process. Lyme and its coinfections incite a hyperresponse of cytokine formation.
Cytokines are proteins made of various types of white blood cells with their function being to make antibodies work more effectively, increase WBC activity, recruit other WBCs to the site of infection, and decrease viral and bacterial replication. Cytokines are produced when infection, oxidizing agents, cytokines, toxins, and other agents stimulate immune cells. Once the immune cells are stimulated, NF-KB causes genetic programming for the production of cytokines and the activation of the leukocytes. With Lyme and other related infections, there is an overproduction of cytokines, inducing an excessive inflammatory response that leads to chronic inflammation. This may manifest itself as a suppressed immune system, pain, reduced hormonal production from the thyroid and adrenal glands, sleep disturbances, cognitive decline, fatigue, myalgias, and depression.6
The dysregulation of cytokines and chemokines is a central feature in the development of neuroinflammation, neurodegeneration, and demyelination in both the central and peripheral nervous systems. This process can lead to activation of the microglia, which may mediate neuronal and glial cell injury and death through the production of the pro-inflammatory factors such as cytokines.
About 15% of the patients with Lyme disease develop peripheral and central nervous system symptoms and involvement. Research indicates that the activation of the inflammatory pathways plays a causal role in the often debilitating and painful symptoms.7
Lyme or Lyme-like diseases can cause arthritis, carditis, and neurologic deficits. When the nervous system is involved, it is called Lyme neuroborreliosis (LNB).8 Microglia and astrocytes are key players in the immune responses within the central nervous system. Studies have shown that microglia and astrocytes express toll-like receptors that play a major role in innate immune responses against microbial pathogens.9 Peripheral nervous system involvement can include facial nerve palsy (Bell's palsy), neurogenic pain radiating along the back into the legs and feet, limb pain, sensory loss, or muscle weakness. Central nervous system involvement may manifest as headache, fatigue, memory loss, learning disability, depression, meningitis, and encephalopathy. Significantly elevated levels of the inflammatory mediators interleukin-6 (IL-6), IL-8, CCL2, and CXCL13 were found as well as increased cell counts of white blood cells in the cerebral spinal fluid.10
Chemokines such as IL-8 and CCL2 are known to mediate the influx of immune cells in the central nervous system in the presence of bacterial meningitis. CXCL12 is known to be the major determinant of B cell recruitment into CSF during neuroinflammation.11 In animal models, upon examination of the dorsal root ganglia, the ganglia showed inflammation with neurodegeneration, along with apoptosis of neuronal and satellite glial cells. Lyme, as a pathogen, also activates the coagulation system by directly turning on the clotting cascade to increase the formation of fibrin leading to hypoxia. This is why the fibrinogen levels may increase in Lyme patients. This process of the activated coagulation pathway is stimulated by the inflammatory cytokines. Some of the pathogens that will activate the inflammatory and coagulation pathway are HHV6, EBV, CMV, Mycoplasma, HS1 and HS2, Chlamydia, Brucellosis, Babesia, Ehrlichia, Bartonella, and Borrelia, many of which are coinfections of Lyme disease. This becomes important when talking about the difficulty in treating these infections and their resistance against treatment. Many bacteria and viruses use fibrin to form protective barriers around themselves, referred to as biofilms or cyst formation. The layer of fibrin covering the microbe makes it almost undetectable by the immune system.12
Biofilms and the Difficulty in Treatment
A biofilm is an accumulation of microbial cells that is irreversibly associated with a surface and enclosed in a matrix of primarily polysaccharide material.13 I usually describe this to my patients as like the goo that is left over after pulling up duct tape. Biofilms are the predominant phenotypes in natural and pathogenic ecosystems. As a pathogenic mechanism, they serve as a population-level virulence factor to provide the infectious organism with virulence traits that a single organism cannot possess. These traits are protective to the invading organism and allow them to persist in the host despite the innate and adaptive immune systems. Biofilms usually are associated with chronicity or persistence – as opposed to acute virulence traits such as toxin production. Most chronic infectious disease processes are associated with biofilm production. The biofilm allows for the infection to attach to the host. The biofilm protects the organism from the host's adaptive immune response, along with being attacked by phagocytes. Biofilms also provide an ideal setting for elevated levels of gene transfer for the invading organism. The gene transfers occur since nearly all of the chronic pathogens that form biofilms contain inducible energy, requiring horizontal gene transfer mechanisms that serve a nonnutritive role, as opposed to using the DNA only as a food source. Horizontal gene transfer (HGT) is defined as the movement of genes between two unrelated cells usually in a unidirectional manner.14 This allows bacteria to transmit genes vertically to daughter cells, or by HGT. Three forms of HGT have been classified: transformation, conjugation, and transduction.
Page 1, 2
|
![]()
![]()
![]()






