|
The Centers for Disease Control and Prevention (CDC) reported nearly 32,500 new cases of Lyme disease in 2011.1 Even though it is estimated that the actual number could be up to 10-fold higher, Lyme disease is still the most reported tick-borne illness in the US.2
There are a variety of reasons for this underreporting, including unclear clinical presentation and limitations of current testing methodologies. Early and accurate diagnosis is important, as patients not receiving proper treatment may develop chronic infection or late-stage Lyme diseases such as chronic Lyme arthritis or chronic Lyme neuroborreliosis, which can be very serious in some cases.3
Lyme disease is caused by B. burgdorferi, a bacterium of the spirochete class. Lyme disease is a zoonotic, vector-borne disease transmitted by the Ixodes (blacklegged) tick. Symptoms generally include some of the following:
- known tick bite
- bull's-eye rash (erythema migrans)
- flu-like symptoms
- lymphadenopathy
- joint pain
- neurological symptoms
- heart palpitations
- severe fatigue
The current CDC-suggested test for diagnosis is a two-tier process including ELISA and western blot (WB) analyses. These tests are serological assays that detect antibodies to B. burgdorferi. The low sensitivity of these tests (about 30% in early Lyme disease and 50% in late Lyme disease) and the significant seronegativity of Lyme patients (as many as 30% to 50% of cases) suggest the need for more sensitive T cell-based laboratory tests.4 The enzyme-linked immunospot (ELISPOT) assay is an effective method for assessment of the magnitude and the quality of T cell immunity by measuring stimulated antigen-specific T cells.5
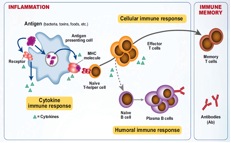
A complete evaluation for Lyme disease requires testing for both a humoral and a cellular immune response (Figure 1). This is done by measuring both antibodies (humoral/WB) and T cell activity (cell-mediated/ELISPOT). A new diagnostic tool for the detection of Lyme disease called iSpot Lyme incorporates both of these pieces and has a sensitivity of 84% and specificity of 94%.
Figure 1
Clinical Utility
The ELISPOT method utilized in iSpot Lyme is a highly sensitive technique for detecting immune cells that secrete signature proteins (such as cytokines). It is the only available technology that accurately detects, measures, and performs functional analysis of low-frequency immune cells. This test detects a cellular immune response against Lyme antigens, which appears earlier in the disease process than the antibody response detected by the traditional western blot test.6 More importantly, iSpot Lyme can detect antigen-specific T cell responses in seronegative patients.7 Therefore, the Lyme ELISPOT test can be used to provide information regarding the current immune status of a Lyme disease patient.
Methodology
The immune response to infection with B. burgdorferi includes both B cell and T cell activation.8 T cells are sensitized to B. burgdorferi antigens and the activated effector T cells produce the cytokine interferon gamma (IFN-g) when stimulated by these antigens.
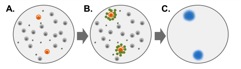
The iSpot Lyme test counts B. burgdorferi-sensitized T cells by capturing interferon-gamma (IFN-g) secreted by these T cells. More specifically, when IFN- g is released, a "spot" of insoluble precipitate is formed at the site of the reaction. Evaluating the number of spot-forming units (SFUs) provides a measurement of B. burgdorferi-sensitive effector/memory T cells in the peripheral blood. The SFU count correlates to a patient's T-cell reaction to B. burgdorferi (Figure 2).
Figure 2
Conclusion
Lyme disease is a serious illness caused by tick transmission of the spirochete B. burgdorferi. Diagnostic criteria have historically relied on serologic testing results based on ELISA and western blot analyses. These tests are limited in the detection of early Lyme infection and have significant potential for false-negative results.
A highly sensitive T cell-based enzyme-linked immunospot (ELISPOT) method, iSpot Lyme can verify exposure to B. burgdorferi antigens. This novel assay complements traditional serologic tests, demonstrates increased sensitivity and specificity compared with previous methodologies, and can increase the speed of diagnosis and treatment, leading to improved clinical outcomes.
iSpot Lyme should be used and interpreted within the entire clinical presentation, and can be useful for patients with clinical indications of Lyme disease who have previously tested negative.

Notes
1. Centers for Disease Control. Reported cases of Lyme disease by year, United States, 2002–2011 [Web page]. http://www.cdc.gov/lyme/stats/chartstables/casesbyyear.html.
2. Yound JD. Underreporting of Lyme disease. N Engl J Med. 1998;338(22):1629–1629.
3. Maloney EL. The need for clinical judgment in the diagnosis and treatment of Lyme disease. J Am Physicians Surg. 2009. 14(3):82–89.
4. Lehmann PV, Zhang W. Unique strengths of ELISPOT for T cell diagnostics. In: Kalyuzhny AE, ed. Handbook of ELISPOT: Methods and Protocols, Methods in Molecular Biology. Vol. 792, 2nd ed. New York: Springer Science+Business Media LLC; 2012:3–23.
5. Tary-Lehmann M, Hamm CD, Lehmann PV. Validating reference samples for comparison in a regulated ELISPOT assay. In: Orabhakar U, Kelley M, eds. Validation of Cell-Based Assays in the GLP Setting: A Practical Guide. 1st ed. West Sussex, UK: John Wiley & Sons Ltd.; 2008:127–146.
6. Ekerfelt C, Forsberg P, Svenvik M, et al. Asymptomatic Borrelia-seropositive individuals display the same incidence of Borrelia-specific interferon-gamma (IFN-g)-secreting cells in blood as patients with clinical Borrelia infection. Clin Exp Immunol. 1999;115:498–502.
7. Dattwyler RJ, Volkman, DJ, Luft, BJ, et al. Seronegative Lyme disease – dissociation of specific t- and b-lymphocyte responses to Borrelia burgdorferi. N Engl J Med. 1988;319(22):1441–1446.
8. Martinuzzi E, Scotto M, Enee E, et al. Serum-free culture medium and IL-7 costimulation increase the sensitivity of ELISPOT detection. J Immunol Meth. 2008;333:61–70.
Dr. Chenggang Jin holds a PhD in immunology and is also an MD with 10 years of experience in medical research at Princeton University, the University of Iowa Hospitals and Clinics, and Beth Israel Deaconess Medical Center/Harvard Medical School. Dr. Jin also holds certification from the Educational Commission for Foreign Medical Graduates in the field of medicine. His specialized training includes many years of technical and research experience in cellular immunology and molecular biology. He also has extensive experience with flow cytometry and PCR instrumentation. He is currently the director of laboratory immunology at Pharmasan Labs and led the creation of a new clinical laboratory assay, iSpot-Lyme, for Lyme disease.
Deanna Fall is currently a scientific writer and research analyst for NeuroScience Inc. and holds a bachelor's degree in biology from Ferris State University in Big Rapids, Michigan. Deanna joined NeuroScience in 2009 with experience in pharmaceutical research and development in the field of neurotoxicology. She is currently pursuing a master's degree in health informatics at the University of Minnesota.
|
![]()
![]()
![]()






