|
Abstract
Two groups of Borrelia, the Borrelia burgdorferi group that causes Lyme disease and the relapsing fever Borrelia group that cause relapsing fever, are found in similar habitats and overlapping locations worldwide. Correct identification of the infecting organism is important because there are related symptoms in the diseases caused by the two groups of Borrelia. We illustrate the diagnostic complexity involved with immunoblotting and PCR and propose that a panel of tests are needed for correctly identifying the two groups of Borrelia.
Introduction
Borreliosis are a group of globally distributed infectious diseases caused by spirochete bacteria of the genus Borrelia that are transmitted by ticks and lice acting as vectors. The two groups of Borrelia known to cause disease in humans are the Borrelia burgdorferi group that causes Lyme disease and the relapsing fever Borrelia group (RFB) that cause relapsing fever. Lyme borreliosis is transmitted by hard ticks (Ixodes species); and RFB are transmitted by the same hard ticks that transmit Lyme disease as well as soft ticks and body lice.
Lyme borreliosis is a disease that can affect many organs including the heart, joints, central nervous system, and brain. Borrelia afzelii causes a distinct skin infection known as acrodermatitis chronica atrophicans (ACA), and B. garinii is associated with early Lyme neuroborreliosis (i.e., painful meningoradiculoneuritis or Bannwarth syndrome). Borrelia burgdorferi sensu lato currently includes at least 18 species of Borrelia world-wide. B. afzelii, B. bavariensis, B. bissettii, B. garinii, B. kurtenbachii, B. burgdorferi sensu stricto, B. spielmanii, B. lusitaniae, and B. valaisiana are found in Europe. B. burgdorferi sensu stricto, B. carolinensis, B. bissettii, B. californensis, B. andersonii, and B. mayoni are found in the USA. The three more important human pathogenic species causing Lyme borreliosis are B. burgdorferi sensu stricto in the USA and Europe; and B. afzelii and B. garinii in Europe and Asia.1
Relapsing fever is also caused by spirochete bacteria of the genus Borrelia, of the group RFB. They are closely related to B. burgdorferi, the causative agent of Lyme disease. RFB are found worldwide and are transmitted by the body lice and ticks. Relapsing fevers can be grouped into louse-borne, soft tick-borne, and hard tick-borne relapsing fevers.
 Patients infected with RFB can get relapsing fevers, severe headache, neck stiffness, arthralgia, myalgia, ecchymosis, epistaxis, and petechiae. Complications include acute respiratory distress syndrome, decreased sensorium, myocarditis, hepatitis, spontaneous abortion or stillborn, and death. However recent studies have shown that infection by RFB can also produce symptoms similar to Lyme disease including fever, headache, fatigue, chills, myalgia, joint and muscle pain, loss of appetite, nausea, disorientation or memory loss, lack of coordination, as well as more severe conditions of neurological disease.2 Patients infected with RFB can get relapsing fevers, severe headache, neck stiffness, arthralgia, myalgia, ecchymosis, epistaxis, and petechiae. Complications include acute respiratory distress syndrome, decreased sensorium, myocarditis, hepatitis, spontaneous abortion or stillborn, and death. However recent studies have shown that infection by RFB can also produce symptoms similar to Lyme disease including fever, headache, fatigue, chills, myalgia, joint and muscle pain, loss of appetite, nausea, disorientation or memory loss, lack of coordination, as well as more severe conditions of neurological disease.2
Louse-borne relapsing fever (LBRF) is caused by a single pathogen, B. recurrentis.3 The first case linked with spirochetal etiology and body louse transmission was documented a century ago. Between 1919 and 1923, 13 million cases resulting in 5 million deaths occurred in Eastern Europe. During World War II, a million cases occurred in North Africa. LBRF is commonly found in Ethiopia, Sudan, Eritrea, and Somalia. Illness can be severe, with mortality of 30% to 70% during disease outbreaks.4 Today, LBRF causes sporadic illness and outbreaks in sub-Saharan Africa, particularly in regions affected by war and in refugee camps.5
Soft tick-borne relapsing fever Borrelia was first discovered to cause disease in humans in the early 20th century. These RFB are found all over the world. Borrelia crocidurae and B. duttoni are found in Africa6, and B. persica, B. latyshervi, and B. hisponica in Asia and the Middle East. Borrelia venezuelensis and B. mazotti are common in Central and South America. In North America, B. hermsii is present in forested mountains at 1,000–2,700 meters particularly in rustic cabins; B. turicatae occurs in desert, semi-desert, scrub landscapes, caves, and rodent burrows while B. parkeri has been recently found in ticks in Florida.7-8
Recently, hard-tick relapsing fever infections have been shown to be caused by B. miyamotoi. Borrelia miyamotoi was discovered in 2005 in Japan.9 In 2011, the first human case of B. miyamotoi infection was reported in Russia.10 In the US, the first case was reported in 2013.11 B. miyamotoi is transmitted by the same hard ticks that transmits B. burgdorferi. Based on this information, it is clear that we need better diagnostic tests to detect and differentiate both groups of Borrelia. We summarize here the progress in developing Western blots and PCR tests for RFB that can help differentiate them from Borrelia species causing Lyme borreliosis.
Methods
Western blot strips were prepared from RFB as previously described by Shah et al.12 Briefly, RFB species were lysed, the lysates subject to sodium dodecyl sulfate-polyacrylamide gel electrophoresis and the proteins then transferred to nitrocellulose membrane for immune-probing with specific rabbit anti-Borrelia antibodies and patient sera in an immunoblotting procedure.
 Real-time PCR was also performed on patient samples using a BioRAD qPCR kit as per the manufacturer's recommendation (Catalog Number 175285, Biorad Laboratories, Hercules, CA). Primers and probes specific for RFBwere designed from published sequences of the 16S ribosomal gene DNA of RFB. Real-time PCR was also performed on patient samples using a BioRAD qPCR kit as per the manufacturer's recommendation (Catalog Number 175285, Biorad Laboratories, Hercules, CA). Primers and probes specific for RFBwere designed from published sequences of the 16S ribosomal gene DNA of RFB.
Findings and Discussion
The western blot results using serum from a patient suspected to have RFB infection when tested against proteins in whole cell lysates from different Borrelia species are shown below in Figure 1. The two blots were developed with alkaline phosphatase-conjugated goat antibodies against human IgM and IgG respectively. The results show the detection of specific protein antigens (blue arrows), by IgM but not IgG antibodies in the patient's serum, in RFB but not in Borrelia species causing Lyme disease, notably B. burgdorferi.
Figure 1: Borrelia Species Western Blot Tested with Serum from a Patient Suspected of Relapsing Fever Borrelia Infection
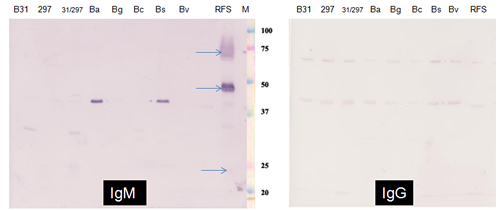
Borrelia burgdorferi Group: B31 - B. burgdorferi B31;
297 – B. burgdorferi 297; Ba – B. afzelii; Bg – B. garinii; Bc – B. californensis; Bs – B. spielmanii; Bv – B. valaisiana
Relapsing Fever Borrelia Group: RFS – Relapsing Fever Borrelia sp.
Several studies have been performed to determine the B. miyamotoi infection rates in field-collected nymphal Ixodes ticks. B. miyamotoi infection rates range between 0–10% in the I. scapularis nymph populations13-17 and between 0–15% in I. pacificus nymph populations.18-21 The reported ratio of B. burgdorferi to B. miyamotoi infection in I. scapularis nymphs in northeastern US range between 4:1 and 6:1. On the West Coast, (1) soft ticks that transmit B. hermsii, and B. turicatae are prevalent; (2) the B. miyamotoi infection rate is between 0%-15% in the I. pacificus populations18-21; and (3) the B. burgdorferi infection rate is lower than on the East Coast.
Consistent with these high infection rates for RFB, we found that more than 50% of the patients who were negative for B. burgdorferi infection based on B. burgdorferi western blot and PCR results had antibodies to RFB; and 9% were also positive for RFB by PCR. Although the clinical sensitivity of RFB PCR is low, it is important to include it as part of testing for the following reasons: (1) 20-30% of the patients never produce antibodies and (2) PCR potentially has the highest specificity for detecting different Borrelia species.
We observed that no patient with Lyme disease-like symptoms that tested positive for RFB remembered having relapsing fevers. Therefore, we propose that patients with Lyme-like symptoms but negative by the B. burgdorferi western blot and PCR tests should be tested for RFB infections. A borreliosis panel that includes both serological and molecular tests for B. burgdorferi group Borrelia and Relapsing Fever Borrelia group appears to be appropriate in this situation.
Acknowledgements
We would like to thank Wai Lo, Akhila Poruri, and Cathy Calalo for technical assistance, and Dr. R. Ramasamy for editing the manuscript.
References .pdf
|
![]()
![]()
![]()
![]()







