Defining
Metal Intoxication
Blood is a part of the human circulatory system. It transports oxygen,
nutrients, and toxins to and from the cells and organs. The nutrients
and toxins taken in will either "feed" body cells, be
stored, or be excreted through the urinary or digestive tract, through
breathing and sweat. While circulating in the bloodstream, toxins
can be measured and monitored, hence the term Human Biomonitoring.
Human monitoring of occupational exposures started in the 1890s
through a variety of blood lead monitoring programs. Population-based
biomonitoring is more recent and has been implemented at various
levels within the United States (both federally and among states)
and internationally. In the US, the recent advent of the National
Health and Nutrition Examination Survey (NHANES) resulted in population-based
biomonitoring studies of lead and cadmium in clinical specimens.
The combined effort, nationally and internationally, improved our
understanding of how widespread some chemical exposures are in the
general population.1
New technology allows the detection of minute amounts of potentially
toxic metals. Never before in the history of medicine have we had
the analytical accuracy to correlate such traces of toxins with
early onset of disease. Modern analytic chemistry allows physicians
to take early action.
Through early diagnosis, we can utilize chelation therapy or metal
detoxification treatments not only for the treatment of acute metal
intoxication, but also for preventive and curative measures. Diagnostic
abilities improve treatment potential. Specific monitoring of low
dose intoxication allows early removal of harmful toxins from the
body to prevent disease. Early intervention enables us to remove
the potential cause(s) of an existing disease.
First Diagnose,
Then Treat
To treat metal intoxication, we must first define the degree of
toxicity. An acute intoxication at the workplace demands another,
more aggressive treatment than a chronic case of metal intoxication.
While a low-level metal exposure can be one cause of chronic diseases
and disease patterns, it is important that we first identify the
type of metal toxicity (lead, mercury, etc.) and the severity thereof.
From that information, we can safely select a) the appropriate chelating
agent and b) the route of delivery and the frequency of treatment.
Diagnosing Acute
Metal Intoxication
Medically, a patient is considered acutely exposed, or toxic, when
his blood levels exceed the Biological Tolerance Value, also referred
to as the BAT level. The BAT value is defined as the maximum permissible
quantity of a chemical substance or its metabolites, or the maximum
permissible deviation from the norm of biological parameters induced
by these substances in exposed humans. The BAT value is established
on the basis of currently available scientific data that indicate
that these concentrations generally do not affect the health of
the employee in any significant adverse way, even when they are
attained regularly under workplace conditions. BAT values are established
on the assumption that persons are exposed at work for at most eight
hours daily and 40 hours weekly. BAT values established on this
basis may also be applied without the use of correction factors
to other patterns of working hours. Interestingly, BAT values are
lower for the German than the US population. A cynic might say that
either Germans are more sensitive than US citizens, or that US BAT
values protect employers more than employees.
BAT values are conceived as ceiling values for healthy individuals.
They are generally established for blood and/or urine and take into
account the effects of the substances and an appropriate safety
margin, being based on occupational medical and toxicological criteria
for the prevention of adverse effects on health.
Whole blood, serum, and urine samples are used as assay materials.
Hair samples are not suitable assay materials for occupational medical
testing, because hair growth is slow and thus the immediate exposure
cannot be verified. Under workplace conditions, the employee's immediate
intoxication is of concern, not the chronic or long-term exposure.
Therefore, in occupational medicine, the diagnosis concerns itself
with immediate intoxication.
Occupational medical treatment is aimed at reducing the toxicity
level to below the BAT level. The most common "treatment"
of acute workplace intoxication is the removal of the patient from
the workplace. Chelation is considered in serious conditions only,
and reported cases are rare. Instead, the patient is monitored via
blood or urine analysis, and as soon as levels fall below the accepted
BAT range (usually within a day or two after exposure), "treatment"
is considered successful. The patient is brought back to the workplace.
In serious cases of intoxication, the patient is voluntarily removed
from the workplace for one week or longer. In debilitating accidents,
a patient is placed into early retirement, and treatment is palliative.
Comparison of Reference Ranges for the Unexposed
and Those Exposed at the Workplace
It is apparent from the data below that, for most metals, a definite
ceiling range does not exist. Different countries and regulatory
agencies provide differing ranges, and these differences are usually
due to the use of various analytical techniques or population models.
For the general population, even for physicians, comparing reference
ranges is cumbersome, because units may be given in mmol/L or µg/dl,
instead of the more common µg/L.
The
Toxicity of (Some) Blood Metals
Arsenic
(Blood): Blood arsenic levels are not considered diagnostically
useful, and the total arsenic concentration may be markedly increased
after dietary consumption of seafood. Urine samples are more valid
for the diagnosis of arsenic intoxication, but these too are influenced
and often rise dramatically after a seafood meal. Hence, when taking
a blood specimen for blood metal testing, the patient should be
instructed not to eat seafood for at least one day prior to sample-taking
and refrain from smoking for as long as possible. Cigarette smoke
does contain arsenic, beryllium, nickel, cadmium, lead, and other
potentially harmful metals. Hair and nail levels are useful only
for diagnosing a past exposure.
Lead (Blood):
Lead in the human body can be measured in blood, urine, bones, teeth,
or hair. By measuring an individual's blood lead level (BLL), we
can detect lead poisoning in adults or children. When blood lead
is high, an increase in erythrocyte protoporphyrin (EP) follows.2
- The standard elevated blood lead level
(BLL) for adults' set by the Centers for Disease Control (CDC)
is 25 micrograms per deciliter (25 µg/dl) of whole blood.
This level recognizes that every adult has accumulated some lead
contamination.
- The level for a child is lower; currently
it is 10 micrograms per deciliter (10 µg/dl) of blood.2
The CDC states that a blood lead level
above 10 µg/dL is a cause for concern. It also states that
lead can impair development even at BLLs below 10 µg/DL.3
The German Environmental Agency's BLLs are lower than those set
by US agencies. (See Table 1.)
In Australia, the acceptable level of lead in blood was lowered
from 25 µg/dL to 10 µg/dL in 1992. In 1993, the National
Health and Medical Research Council (NH&MRC) set a national
target for 1998 for all Australian to have a BLL less than 15 µg/dL
(except where they worked with lead), and strategies were put in
place whereby 90% of pre-school children would have BLLs below 15
µg/dL. In 1996, the National Blood Lead Survey (the Donovan
Survey) found 7.7% of children aged one to four were above 10 µg/dL,
and 1.7% were above 15 µg/dL.4
Biomonitoring Ranges for a Normal, i.e., Non-Exposed,
Population; Levels Above the Given Range Indicate Need for Action:
Children (USA)
Adults (USA)
Children and adults (Germany)
Females (18-69yrs) (Germany)
Males (18-69yrs) (Germany) |
< 100 µg/L = 10µg/dl
<250
µg/L
<50 (Table 1)
<70 (Table 1)
<90
(Table 1) |
- CDC recommends that all children be
screened for lead poisoning yearly. This is especially important
for children between six months and six years of age.
- Children with an erythrocyte protoporphyrin
level (EP) of 35 micrograms per deciliter (=350µg/l) should
be tested for a blood lead level.
- Children with a BLL of 20 micrograms
per deciliter (=200µg/L) or higher should be screened by
their doctor for lead poisoning.
- Medical treatment is necessary if
the BLL is higher than 45 micrograms per decilitre (=450 µg/l).
Levels of Acute
Exposure as Utilized in Occupational Medicine
Adults (occupational exposure US ranges)
OSHA action level
BEI (Biological Exposure)
BAT (Biological Tolerance) |
>40 µg/dl = 400µg/L
>30
µg/dl = 300µg/L
>70 µg/dl = 700µg/L |
The OSHA Safety and Health Achievement
Recognition Program (SHARP) collect and maintain a registry of blood
lead levels by occupation and industry. Table 1 shows elevated blood
lead levels measured in Washington State construction workers. Blood
results are reported in micrograms per deciliter (µg/dl),
and the data shows that exposure is common. Companies that do not
test their workers are not represented, and sadly, enough many exposed
workers do not have their blood tested.2 (Note: 1mcg/dl = 10mcg/L)
Table 1: Blood
Levels in Washington State Construction Workers (100KB
.pdf)
Treatment Options
All forms of EDTA (NaEDTA, NaMgEDTA, CaEDTA) have a high lead-binding
capacity. CaEDTA has been approved by the FDA to chelate lead, and
the proper infusion rate is 1gr/hr. If infused too quickly, EDTA
is nephrotoxic. Although CaEDTA bolus injections are becoming increasingly
popular, it is dangerous to administer EDTA at such a fast rate.
The International Board of Clinical Metal Toxicology (IBCMT) strongly
advises against it.7
Cadmium (Blood):
According to the Agency for Toxic Substances and Disease
Registry (ATSDR), elevated blood cadmium levels confirm acute exposure,
(Jarup 2002; ATSDR 1999) but do not correlate with body burden or
clinical outcome.5 According to the ATSDR, a blood test alone is
not sufficient validation for treatment, possibly because blood
cadmium levels are easily influenced through smoking or smoke exposure.
The 95% confidence limit for blood cadmium levels in the United
States for healthy nonexposed, nonsmokers is 0.4 micrograms per
liter (µg/L) (CDC 2005). ATSDR recognizes that occupationally
exposed persons may have higher blood levels than the general population.
OSHA (www.osha.gov) considers a whole blood level of 5µg/l
or higher hazardous.5
German agencies have set stricter standards:
Non-smoking children 6-12 years <0.5µg/L
Non-smoking adults, 18-69 years <1.0µg/L
Treatment Options
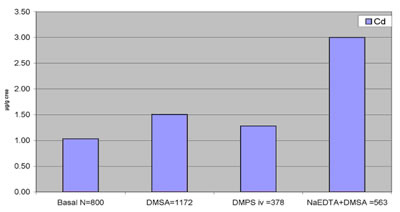
All forms of EDTA (NaEDTA, NaMgEDTA, CaEDTA) bind cadmium. A recent
statistical evaluation by Micro Trace Minerals Laboratory of post
chelation urine tests (Table 2) indicates that EDTA seems to be
the best option available at this time.
Table 2: Cadmium-Binding
and Urine Excretion After Various Chelation Treatments
Source Micro Trace Minerals, Germany/Boulder,
Colorado
Mercury (blood):
Blood levels are used as markers to determine the severity of a
mercury exposure. For standard blood mercury test, mercury is measured
as total mercury (inorganic and organic). Except for methylmercury
exposures, blood is considered useful if samples are taken within
a few days of exposure. This is because most forms of mercury in
the blood decrease by one-half every three days if exposure has
been stopped. Thus, mercury levels in the blood provide more useful
information after recent exposures than after long-term exposures.6
Human Biomonitoring
Range:
Normal (unexposed population): < 5.8 µg/L (US Environmental
Protection Agency)
Normal (unexposed population): < 8 µg/L (University of
Iowa, USA)
Normal (unexposed population): < 2 µg/L (Environmental
Protection Agency, Germany)
Levels of Acute
Exposure as Utilized in Occupational Medicine:
BEI (Biological Exposure Index): 15 µg/L (total inorganic,
end of shift, end of work week)
BAT (Biological Tolerance Value): 50µg/L (organic and inorganic)
BAT (Biological Tolerance Value):100µg/L (organic)
Treatment Options
When a blood test no longer reflects a mercury exposure, a DMPS
challenge test may be performed to confirm or rule out mercury intoxication.
DMSA can also be used to provoke mercury binding and excretion,
though the binding ability of intravenously administered DMPS is
much stronger.
Note: many physicians assume that
oral DMPS has the same binding capacity as IV DMPS. The fact is,
the mercury-binding ability of oral DMSA and oral DMPS is similar,
and both of these oral chelators have a lower mercury binding capability
than IV DMPS.
Blood Sampling
Specifics
Collection Medium: Metal
free royal blue EDTA tube
Please note that Heparin or regular EDTA tubes are no longer used
for metal testing due to contamination.
Minimum:
3 mL whole blood
Analysis:
Must be AA hydrid method or ICP-MS with collision or cell reaction
technique. All other ICP-MS instruments are too much affected by
interferences and are unable to have the needed sensitivity to detect
low levels. False highs may be a problem.
Analytical Time:
Two days to one week
Diagnosing Chronic
(Over)Exposure
Biomonitoring ranges apply for a population considered "nonexposed."
Ironically and sadly, the long-term exposed are often chronically
ill people with sad histories of "unknown cause." Medically,
they are considered "unexposed" until the diagnosis indicates
a blood or urine value above the biomonitoring range. Table 3 shows
human biomonitoring ranges as set by the German Environmental Agency.
These ranges apply to people not working in industries that may
lead to occupational exposure. People with past exposures, or those
exposed to low levels on a daily basis, may or may not show blood
levels above these ranges. Most importantly, unremarkable results
do not rule out chronic exposure.
Table
3: Biomonitoring Ranges Last Updated by German Environmental Agency
2005 (528KB .pdf)
When we suspect past or chronic exposure,
but blood tests are negative, we consider a "challenge test,"
also referred to a "provocation test." By introducing
a chelating substance into the bloodstream, we force metal binding
and excretion. Results are often astonishing. Depending on how much
of a metal has been stored in the body, urine excretion levels may
rise well above the expected range. In most cases, patients respond
favorably, if not unexpectedly. Symptoms, even unrelated ones, may
disappear. Every doctor practicing chelation therapy has such case
histories.
Case History:
Beate, a 45-year-old biologist, works in our laboratory. She is
extremely disciplined and efficient, but rheumatoid arthritis (with
a high positive RA factor) has proven to be a challenge since her
early twenties. She had been on cortisone, but stopped after experiencing
strong side effects. During her frequent rheumatic attacks, strong
pain medication was her only alternative. Beate has suffered from
asthma since childhood and also suffers from Hashimoto disease.
She currently takes thyroxin, 175 mcg daily. During history taking,
it became apparent that the Hashimoto appeared after the removal
of her many amalgam fillings, which her dentist took out "all
at once and without any precautions." At that time, she experienced
multiple food sensitivities and an allergy to penicillin. Migraine
became another problem.
Mercury overexposure seemed a reasonable diagnosis. Hair mercury
levels were at 1.89mg/kg (=ppm), far exceeding the upper range of
0.6 ppm. Medical analysis showed a normal renal function and blood
pressure. We first tested her reaction to DMSA by giving her 500
mg under close supervision. Her urine mercury excretion level was
a modest 3.37 mcg/g creatinine. Other than feeling weak and light-headed,
she noticed no side effects. The next day, she felt amazingly well.
Joint swelling and pain was noticeably decreased.
We supported her nutritionally before the next "challenge test"
two weeks later. Again, she felt weak and light-headed, urine results
showed a slight increase in urine mercury at 5.36µg/g crea,
and again, the day after, she was without pain and felt energetic.
We have continued this treatment cycle, and so far, results have
been amazing. After three months of biweekly treatment, DMSA was
increased to 1000 mg and urine mercury excretion rose to a significant
36.2µg/g crea. She continued to be symptom-free. A repeat
RA factor turned out to be normal. Two weeks later, she experienced
a monosodium glutamate (MSG) reaction after eating at an Italian
restaurant. A severe migraine was followed by another rheumatic
attack.
The treatment cycle was temporarily stopped.
Choosing the Most
Appropriate Chelator
Why did we, in Beate's case, not use IV DMPS, which is a stronger
mercury chelator? We didn't do so for two reasons:
1. Beate is hypersensitive and afraid
of experiencing reactions. A softer approach seemed warranted.
2. DMPS injectables were unavailable at the time. We could have
used oral DMPS, but it has a similar binding as DMSA. We did not
use oral DMPS, because it has a stronger affinity to bind zinc,
and Beate's hair analysis showed borderline zinc levels. Unlike
oral DMPS, DMSA does not bind zinc in any significant way.
Every chelating agent has a specific
binding capacity to certain metals, and we can enhance the effectiveness
of chelation therapy by paying attention to those chemical specificities.
Similarly, we reduce the chelation benefit by ignoring "finer
points."
Diagnostically, it is important to find
out the type and severity of the existing metal intoxication. In
addition, it is important to identify existing deficiencies and
pay attention to borderline deficiencies. If we would use DMPS (or
EDTA) on a borderline zinc-deficient patient, we could create an
acute deficiency. Consequently, we must initiate a nutritional program
before chelation is started to prevent potential problems. Zinc
deficiency symptoms are not unknown among patients who have undergone
chelation therapy and who have experienced side effects after DMPS
or EDTA treatment. A proper supplementation schedule could have
avoided the problems.
Before any chelation treatment is started, we must know renal function
and order additional diagnostic tests, depending on the patient's
health problems. A cardiac patient will require a different diagnostic
schedule than a neurological patient. After we diagnostically defined
the patient's general health status, we can select the appropriate
and most effective chelating agent.
Are So-Called Unexposed
Patients in Need of Chelation?
The term "unexposed" is used for people who do not work
in a hazardous working environment and have not been exposed to
environmental- or industry-related accidents. Unfortunately, chronic
metal intoxication exists more than we are willing to admit. The
following excerpt of the New York City
Health Report of July 23, 2007 should be a warning. It indicates
that one in four New Yorkers has elevated blood mercury levels,
a clear sign of mercury overexposure.
Today's findings are the latest presented from New York City's Health
and Nutrition Examination Survey (NYC-HANES), the first such survey
ever conducted by a US city. It's possible that other cities have
similarly high levels, or higher ones, but haven't yet documented
them. Because mercury is a concern for the health of newborns, recommendations
on mercury exposure are most important for pregnant and breastfeeding
women.
- Among women 20-49 years old in New
York City, the average blood mercury level is 2.64 µg/L
(micrograms per liter), three times that of similarly-aged women
nationally (0.83 µg/L).
- Approximately one-quarter of New York
City women in this age group have a blood mercury level at or
above 5 µg/L, the New York State reportable level.
- People who eat fish three or fewer
times each week have, on average, levels of mercury below the
reportable level, while average readings exceed the reportable
level among those who eat fish four or more times.
- Higher-income New Yorkers have higher
mercury levels; New Yorkers in the highest income bracket average
3.6 µg/L, compared to 2.4 µg/L among the lowest income
group.
- Average blood mercury levels are considerably
higher among New York City Asian women (4.1 µg/L); nearly
half (45%) have blood mercury levels at or above the State reportable
level.
- Among Asians, foreign-born Chinese
women have particularly high levels compared to the rest of New
York City. Two-thirds (66%) have mercury at or above the reportable
level.
- Foreign-born Chinese New Yorkers eat
an average of three fish meals per week, compared to about one
among New Yorkers overall. About one-quarter of Chinese New Yorkers
eat fish five or more times each week, compared to fewer than
one in 15 overall.
Should we routinely check whole blood
metals? Do one in four New Yorkers need chelation? The New
York City Health Report speaks for itself. Is the mercury
problem unique to New Yorkers? It would be naïve to believe
that.

Notes
1. Committee on Human Biomonitoring for Environmental Toxicants,
National Research Council; Human Biomonitoring for Environmental
Chemicals (2006), Board on Environmental Studies and Toxicology
(BEST)
2. Dept. of Ecology, State Washington. Available at: http://www.ecy.wa.gov/programs/hwtr/demodebris/pages2/lbloodtest.html
3. CDC Weekly. December 22, 2000;49(50):1133-7.
Available at: www.cdc.gov/mmwr/preview/mmwrhtml/mm4950a3.htm
4. Australian Government. Dept. of Environment, Water, Heritage
and the Arts. Available at: http://www.deh.gov.au/settlements/chemicals/index.html.
5. Agency for Toxic Substances and Disease Registry (ATSDR). 1999.
Toxicological profile for Cadmium. Atlanta, GA: U.S. Department
of Health and Human Services, Public Health Service.
6. Agency for Toxic Substances and Disease Registry (ATSDR). 1999.
Toxicological profile for Mercury. Atlanta, GA: U.S. Department
of Health and Human Services, Public Health Service.
7. Van der Schaar P. IBCMT Textbook of
Clinical Metal Toxicology. 2008. Available at: www.ibcmt.com.
|



![]()
![]()
![]()


