|
Lyme disease was first described in 1975 as "Lyme arthritis" in a group of patients with coexisting symptoms and later renamed "Lyme disease."1 Whatever its name, Lyme is currently the fastest-growing vector-borne disease. The Centers for Disease Control and Prevention (CDC) reported nearly 32,500 new cases in 2011.2 However, it is estimated that the actual number could be up to 10 times higher.3 This makes  Lyme disease an epidemic larger than AIDS, West Nile virus, and Avian flu combined.4-6 Lyme disease an epidemic larger than AIDS, West Nile virus, and Avian flu combined.4-6
Lyme disease is caused by Borrelia burgdorferi, a spirochete transmitted as a zoonotic disease by the Ixodes scapularis (black-legged) tick. Fewer than half of all patients recall having been bitten, and only about three-fourths of infected patients develop the trademark bull's-eye rash. Complicating the diagnosis of Lyme disease is its clinical symptomatology, which mimics that of many common disease states and infections (see Figure 1). These may include:
- fatigue
- achy joints/pain/stiffness
- cognitive issues
- headaches/migraines
- tingling/numbness/shooting pains
- shortness of breath
- arthritis
- fibromyalgia
- heart palpitations
Atypical presentation and inaccurate testing frequently lead to underreporting.3 Patients not receiving adequate treatment may develop chronic infection or late-stage Lyme diseases, such as chronic Lyme arthritis or chronic Lyme neuroborreliosis, which can be devastating in some cases.7
Figure 1
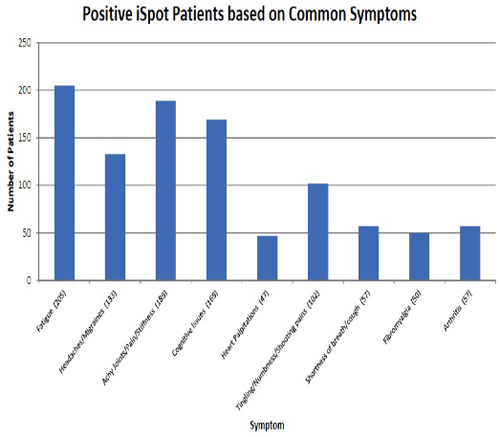
Meta-analysis performed by Pharmasan Labs 2014 (submitted for publication).
Many common complaints can accompany Lyme disease, including fatigue, achy joints, cognitive issues, and headaches.
Case Study
During my internal medicine residency in Arizona, two brothers, aged 24 and 22, arrived together at the emergency room. The men were seemingly healthy, on no medications, and having no significant medical history. They reported that soon after a recent three-week canoeing trip in Minnesota, they each began experiencing fever and arthritislike symptoms. The joint pain was asymmetric and seemed to migrate from one joint to the other.
All autoimmune and inflammatory markers were negative. Chemistry and blood counts were normal, with the exception of the younger brother, who had a slightly elevated platelet count. He reported that on the second day of his trip, he had observed a tick on his skin, which he subsequently removed. Still, a bull's-eye rash appeared in that area the next day.
Both brothers were negative on ELISA and western blot for an antibody response to B. burgdorferi. Based on the younger brother's report of the tick, the bulls-eye rash, and elevated platelets, tick-based illness was diagnosed. The younger brother was prescribed doxycycline 100 mg twice per day for 14 days. Ultimately he made a full recovery.
Back at the ER, the older brother reported joint pain but did not recall a tick bite. Unlike his younger brother, he had no distinctive rash and showed no elevated platelets. Thus he was released without treatment.
Fast-forward 3 years, when I was doing rounds in the hospital's neurology clinic. There, I was surprised to again see the older brother, who was being evaluated for multiple sclerosis (MS). The brother told me that periodically over the past year he had been experiencing double vision and vertigo, with numbness and tingling in both arms, and was having trouble with short-term memory and concentration.
He had previously undergone an EMG of his upper extremities and a full autoimmune workup (due to a family history of lupus), and all labs and chemistries came back normal. He did not meet clinical criteria for MS, although he did have an abnormal brain MRI, which did not correlate to his clinical symptoms.
Recalling the Minnesota canoe trip 3 years prior, I suggested that, like his younger brother, a tick bite could be causing his perplexing symptoms. I explained that ticks are often smaller than a poppy seed and that many people never recall a bite or display a rash. As a result, the patient did seek out a physician trained in chronic Lyme. After undergoing the ILADS treatment protocol of 1 year of antimicrobial therapy, the older brother ultimately joined his younger brother in a return to full health.
Evaluating Lyme Disease
The current CDC-recommended evaluation for diagnosis of Lyme is a two-tier testing protocol, including both ELISA and western blot analyses. These tests are serological assays that detect antibodies to B. burgdorferi. Such tests (known as B cell-based or humoral tests) are often not sensitive enough to detect the antibody response (as in the brothers' cases), or do not detect it quickly enough to prevent chronic infection.
This two-tier testing method has very low sensitivity and specificity, with a significant number of false positive and negative tests. The high false-negative rates are of particular concern, as many Lyme patients are left undiagnosed and untreated. Due to these factors, many Lyme specialists believe that more sensitive T cell-based laboratory tests should be developed.8
The ELISPOT Assay
One such T cell-based test is the enzyme-linked immunosorbent spot (ELISPOT) assay. Newly available in a test called iSpot Lyme, this test is showing promise as a new, innovative method for assessment of the magnitude and quality of T cell immunity through the measurement of stimulated antigen-specific T cells.9,10 Requiring a blood sample, iSpot Lyme was developed by Pharmasan Labs, a CLIA-approved lab with expertise in testing for tick-borne diseases.
The test uses a method already approved and sanctioned by the FDA and CDC for assessing a tuberculosis infection and simply applies it to the Lyme bacteria B. burgdorferi. While other tests are predicated on a B cell antibody response, the iSpot Lyme test is unique in that it examines the primary mediator of the cell-mediated branch of the immune system: the T cells.
As such, it is a highly sensitive technique for detecting immune cells that secrete signature proteins (such as a given cytokine). With a sensitivity of 84% and a specificity of 94% for the detection of B. burgdorferi, it is the most sensitive technology currently available for detection, measurement, and functional analysis of these immune cells. Thus, it is an excellent complement to the current two-tiered antibody methods.
Methodology
The ELISPOT method utilized in iSpot Lyme detects a cellular immune response against Lyme antigens, which appears 4 to 6 days into the disease process, as compared with 4 to 6 weeks for the antibody response detected by western blot. Perhaps more importantly, this method can detect antigen-specific T cell responses in seronegative patients.11 Therefore, the test can be used to provide information regarding the current immune status of a Lyme disease patient.
Test results are produced by measuring interferon-gamma (IFN-g), which is secreted by T cells in response to stimulation by the B. burgdorferi antigens DbpA, PspC, p100, and VisE-1. Put another way, if a patient has been previously exposed to B. burgdorferi, his or her circulating T cells will release IFN-g when T cells are challenged with the Borrelia antigens.
The test counts B. burgdorferi-sensitized T cells by capturing IFN-g secreted by these cells. When IFN- g is released, a "spot" of insoluble precipitate is formed at the site of the reaction. Evaluating the number of spot-forming units (SFUs) provides a measurement of B. burgdorferi-sensitive effector/memory T cells in the peripheral blood. The SFU count correlates to a patient's T cell reaction to B. burgdorferi. (See Figure 2)
Figure 2
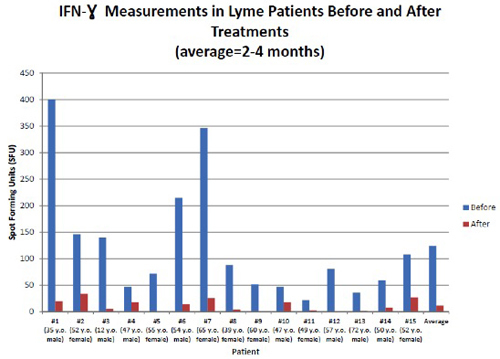
Meta-analysis performed by Pharmasan Labs 2014 (submitted for publication).
The high specificity of the IFN-gamma response as measured in iSpot Lyme spot-forming units (SFUs) is shown here in a group of confirmed Lyme disease patients pre- and posttreatment.
Conclusion
Most clinicians will utilize multiple testing methodologies in complex clinical presentations, and Lyme disease is no exception. My recommendation is to use the cell-mediated ELISPOT technology utilized in iSpot Lyme to identify the highly specific, T cell-mediated activity, combined with the traditional humoral antibody response of a western blot. This provides a two-pronged approach to reduce false negatives.
A study published in Cells, a peer-reviewed, international, open-access journal, validated iSpot Lyme's T cell-based assay and was selected as a "Feature Paper of 2013" for its significant contribution to the science of cell biology.12
As I think back to my experience with the two brothers returning from a Minnesota canoe trip, I realize that the addition of the iSpot Lyme test during the clinical workup could have made a huge difference. It may have provided the additional information needed to provide proper treatment early in the infection, avoiding unnecessary pain and suffering for both of the men.
Notes
1. Arthritis and Lyme disease [online article]. WebMD. http://www.webmd.com/rheumatoid-arthritis/arthritis-lyme-disease.
2. Reported cases of Lyme disease by year, United States, 2003-2012 [Web page ]. CDC. http://www.cdc.gov/lyme/stats/chartstables/casesbyyear.html.
3. Young JD. Underreporting of Lyme disease. N Engl J Med. 1998;338(22):1629–1629.
4. Statistics overview [Web page]. CDC. http://www.cdc.gov/lyme/stats/chartstables/casesbyyear.html.
5. West Nile virus [Web page]. CDC. http://www.cdc.gov/westnile/index.html.
6. Cumulative number of confirmed human cases for avian influenza A(H5N1) reported to WHO, 2003-2012 [Web page]. World Health Organization. http://www.who.int/influenza/human_animal_interface/EN_GIP_20121217CumulativeNumber
H5N1cases.pdf.
7. Maloney EL. The need for clinical judgment in the diagnosis and treatment of Lyme disease. J Am Physicians Surg. 2009;14(3):82–89.
8. Lehmann PV, Zhang W. Unique strengths of ELISPOT for T cell diagnostics. In: Kalyuzhny AE, ed. Handbook of ELISPOT: Methods and Protocols. Methods in Molecular Biology, vol. 792. 2nd ed. New York: Springer; 2012:3–23.
9. T-Spot. TB 9. Package insert. Oxfordshire, UK: Oxford Immunotec Limited; 2009.
10. Tary-Lehmann M, Hamm, CD, Lehmann, PV. Validating reference samples for comparison in a regulated ELISPOT assay. In: Orabhakar U, Kelley M, eds. Validation of Cell-Based Assays in the GLP Setting: A Practical Guide. 1st ed. West Sussex, UK: John Wiley & Sons Ltd; 2008:127–146.
11. Dattwyler RJ, Volkman DJ, Luft BJ, et al. Seronegative Lyme disease- dissociation of specific T- and B-lymphocyte responses to Borrelia burgdorferi. N Engl J Med. 1988;319(22):1441–1446.
12. Jin C, Roen DR, Lehmann PV, Kellermann GH. An enhanced ELISPOT assay for sensitive detection of antigen-specific T cell responses to Borrelia burgdorferi. Cells. 2013;2(3):607–620. doi:10.3390/cells2030607. Available at http://www.mdpi.com/2073-4409/2/3/607.
 With more than 25 years of health-care experience, Dr. Mark Menolascino completed medical school at the University of Nebraska Medical Center, followed by board certification in internal medicine at Banner Health. He holds additional board certifications in holistic medicine, advanced hormone management, and anti-aging medicine, and earned a master's degree in pharmacology and immunology. Dr. Menolascino has advanced training and clinical experience in nutrition, naturopathic, Chinese medicine/acupuncture, Ayurvedic medicine, and homeopathy. In addition to treating patients at his clinical practice in Jackson Hole, Wyoming, Dr. Menolascino serves as medical director for NeuroScience Inc., which provides health-care professionals with integrative clinical assessments and proprietary nutraceuticals to deliver personalized treatment options. With more than 25 years of health-care experience, Dr. Mark Menolascino completed medical school at the University of Nebraska Medical Center, followed by board certification in internal medicine at Banner Health. He holds additional board certifications in holistic medicine, advanced hormone management, and anti-aging medicine, and earned a master's degree in pharmacology and immunology. Dr. Menolascino has advanced training and clinical experience in nutrition, naturopathic, Chinese medicine/acupuncture, Ayurvedic medicine, and homeopathy. In addition to treating patients at his clinical practice in Jackson Hole, Wyoming, Dr. Menolascino serves as medical director for NeuroScience Inc., which provides health-care professionals with integrative clinical assessments and proprietary nutraceuticals to deliver personalized treatment options.
|
![]()
![]()





 Lyme disease an epidemic larger than AIDS, West Nile virus, and Avian flu combined.4-6
Lyme disease an epidemic larger than AIDS, West Nile virus, and Avian flu combined.4-6 

 With more than 25 years of health-care experience, Dr. Mark Menolascino completed medical school at the University of Nebraska Medical Center, followed by board certification in internal medicine at Banner Health. He holds additional board certifications in holistic medicine, advanced hormone management, and anti-aging medicine, and earned a master's degree in pharmacology and immunology. Dr. Menolascino has advanced training and clinical experience in nutrition, naturopathic, Chinese medicine/acupuncture, Ayurvedic medicine, and homeopathy. In addition to treating patients at his clinical practice in Jackson Hole, Wyoming, Dr. Menolascino serves as medical director for NeuroScience Inc., which provides health-care professionals with integrative clinical assessments and proprietary nutraceuticals to deliver personalized treatment options.
With more than 25 years of health-care experience, Dr. Mark Menolascino completed medical school at the University of Nebraska Medical Center, followed by board certification in internal medicine at Banner Health. He holds additional board certifications in holistic medicine, advanced hormone management, and anti-aging medicine, and earned a master's degree in pharmacology and immunology. Dr. Menolascino has advanced training and clinical experience in nutrition, naturopathic, Chinese medicine/acupuncture, Ayurvedic medicine, and homeopathy. In addition to treating patients at his clinical practice in Jackson Hole, Wyoming, Dr. Menolascino serves as medical director for NeuroScience Inc., which provides health-care professionals with integrative clinical assessments and proprietary nutraceuticals to deliver personalized treatment options.