|
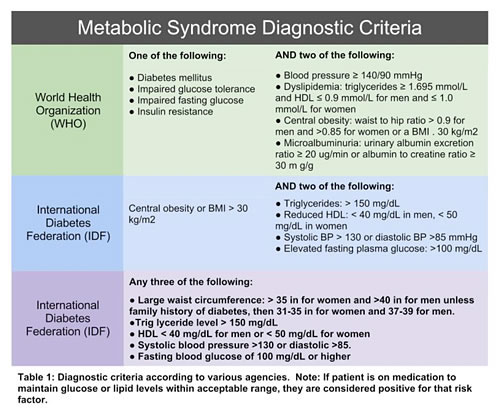
Over the past several decades, metabolic syndrome has become so commonplace that it almost needs no introduction. A CDC report from 2009 estimates that 34.4% of Americans meet the criteria for metabolic syndrome, and this prevalence increases with age (51.5% of males over age 60 fell into this category).1 A "diagnosis" of metabolic syndrome varies slightly depending on whom you ask; however, most are looking at similar symptoms and an increase in mortality has been seen when one or two of these signs and symptoms are exhibited.2
In addition to these common benchmarks of cardiovascular and metabolic health, there are additional laboratory values that may be considered markers of metabolic syndrome, including elevations in C-reactive protein, fasting insulin levels, homocysteine, and, in male patients, low or suboptimal testosterone levels.
Testosterone is certainly recognized as an important aspect of men's health, although many patients and physicians only consider evaluating levels when there is sexual dysfunction present. In fact, the presence of erectile dysfunction is often the symptom that prompts evaluation and treatment that can reduce cardiovascular risk and improve insulin utility.3-5 Fortunately, more men are seeking medical attention. Over the past few decades, we have seen a shift in media portrayal from stoic John Wayne types who rarely admitted to any health concern (let alone sexual dysfunction) to frequent commercials advertising for medications that promise on-demand and lasting erections, and the use of prominent politicians and football personalities as spokesmen for pharmaceuticals and OTC men's health products.
The "little blue pill" may not address the primary problem for most men, but it has played an important role in men's health, if only by prompting important discussion between patients and physicians. So, how common is low testosterone? What are the consequences and symptoms associated with it? How does testosterone affect cardiovascular health and insulin utility? What is the best way to evaluate testosterone levels? What can be done about it? These are important questions, as they pertain to all male patients, particularly those with known risk for metabolic syndrome and cardiovascular disease.
Low or suboptimal testosterone levels in men are more common than many people think. A study in 2006 designed specifically to estimate the prevalence of hypogonadism looked at over 2000 patients and concluded that approximately 38.7% of men over age 45 had total testosterone levels below 300 ng/dl.6 This is a significant number on its own, but the situation may be even worse when accounting for the fact that the majority of the hormone is bound to carrier proteins including sex hormone binding globulin (SHBG) and albumin and, as such, may not be available for tissues to use.
Total testosterone (the value measured in this study) refers to the fraction of the hormone that is bound to carrier proteins as well as the portion that is unbound and available to tissues. Testosterone, like all steroid hormones, is carried in the bloodstream by carrier proteins because it is a lipophilic (fat-loving) molecule that doesn't fare well in the aqueous environment of the blood serum. When the hormone is bound to the carrier proteins (including SHBG, albumin, and others), it is unable to bind to hormone receptors at the tissue level and therefore ineffective in its action. Approximately 95% to 98% of testosterone is bound to a carrier protein at any given time, leaving just the remaining 2% to 5% as completely unbound and available for tissues to use. Because albumin binds to testosterone with a lesser affinity than SHBG, the fraction of the hormone that is bound to albumin is partially available to tissues so the situation isn't quite as "black and white" as it is often presented. This is an important concept to understand when evaluating what various tests are telling us about hormone levels.
In addition to a total testosterone level, most serum laboratories offer a free testosterone level, which is a calculated value based on SHBG levels or determined with equilibrium dialysis to evaluate the percentage of the total level that is unbound. Some laboratories may also offer a value for bioavailable testosterone, which includes both the free fraction as well as the portion of the hormone that is bound to albumin. In addition to serum analysis, there are other ways to effectively measure hormone levels. Salivary hormone levels represent the bioavailable fraction of hormone (both unbound and weakly bound to albumin) because the hormone enters the salivary gland by passive diffusion just as it does other tissues in the body.7 Saliva is not an effective way to measure many body chemistry markers; however, the presence of nonpolar molecules (such as the steroidal hormones) in saliva is non-gradient dependent and directly correlated with the hormone that is available to tissues, making it a more direct measure of bioavailable hormone than serum levels.8
Many studies on hypogonadism as well as many clinicians use a total testosterone level to gauge whether a patient or study participant has low testosterone. If total levels are low, there is a high likelihood that the amount of hormone available to target tissues is diminished; however, there may be many cases wherein total levels do not fall below the reference range, yet the amount available for action is inadequate. This discrepancy in what is tested and what the body has access to often contributes to a large population of men who are subclinical or symptomatic yet go untreated. A more sensitive test would be salivary testosterone levels or free testosterone in serum.
Testosterone has a known age-related decline, and total levels typically drop by approximately 1.6% per year beginning for most men in their 30s. A portion of the testosterone that is produced is converted into estrogen via the aromatase enzyme, and a number of factors increase aromatase activity, including weight gain and environmental exposures. As estrogen levels rise, they prompt the body to produce more SHBG, which in turn has a higher binding affinity for testosterone, and drives the unbound fraction of the testosterone pool down even further.9 When the increase in SHBG is taken into account, the age-related decline in the level of hormone that can be used by the body is closer to 2% to 3% per year.10 Additional factors that influence testosterone levels include stress, history of infection, mechanical trauma, diet, alcohol use, and environmental endocrine disruptors.11-13
Limiting these exacerbating influences is important to incorporate into the treatment plan for men of all ages, and explains why we sometimes see inadequate testosterone levels in young men. The symptoms of low or suboptimal testosterone levels are varied. Many men experience hot flashes, decreased mental clarity, reduced stamina, increased apathy, sexual dysfunction, depressed feelings, muscle aches or joint pain, loss of bone density, and signs of rapid aging as well as increased abdominal obesity, hypertension, and blood sugar imbalances.
The relationship between testosterone and insulin utility is one of mutual causation; that is, a change in one affects the other and vice versa. Men who have been treated with androgen-suppressive therapies have developed insulin resistance, and increased insulin resistance and concomitant increase in body fat percentage lead to a reduction in testosterone both through enhanced aromatase activity and increased leptin production. Leptin, an adipose-derived peptide hormone that regulates appetite and metabolism, has been shown to directly inhibit testosterone production in animal models.14-16 Lowered testosterone levels in turn contribute to additional weight gain and worsening insulin resistance, and so on. As with the chicken and the egg, it is difficult to know which comes first. What we do know is that there is an inverse relationship between testosterone level and insulin sensitivity and that these problems can propagate each other, leaving many men in a downward spiral. There are a number of ways that testosterone affects insulin sensitivity and cardiovascular health (see Figure 1), and much of it is mediated through fluctuations in visceral adipose tissue.
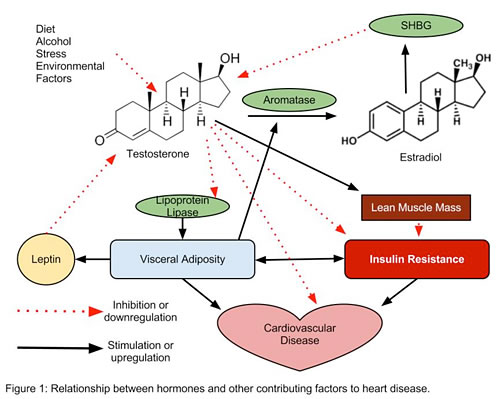
- Fat deposition around the organs as evaluated with MRI and CT scans has a much higher correlation with insulin resistance than subcutaneous adipose tissue.17 Testosterone reduces lipoprotein lipase (LPL) activity and associated uptake of triglycerides into visceral adipose cells, thereby reducing the size of these highly insulin resistant cells.18,19
- Testosterone is an anabolic hormone that helps to increase lean muscle mass and is known to improve insulin sensitivity in muscle cells of rats.20,21
- Testosterone replacement in hypogonadal men may also alter cytokine profiles, shifting from a pro- to anti-inflammatory state, which is important because inflammation is a significant contributor to cardiovascular disease. Additionally, inflammatory cytokines including tumor necrosis factor alpha (TNF-alpha) and interleukin-1 (IL-1) further inhibit Leydig cell testosterone production.22,23
Bottom line: Regardless of elucidated mechanism, it has repeatedly been shown that increasing testosterone levels improves insulin resistance and abdominal obesity, and reduces risk of cardiovascular disease; therefore, boosting testosterone levels and is often an effective way to intervene and shift momentum.24-28
Testosterone replacement therapy, as with most hormone replacement therapies, remains somewhat controversial, and many theories and protocols are used. One longstanding concern about using testosterone replacement in hypogonadal men is that of triggering prostate cancer. Though monitoring of prostate specific antigen (PSA) and prostate size with digital rectal exam (DRE) or ultrasound is recommended for men on testosterone replacement therapy, a significant number of studies published over the past decade are dispelling the myth that testosterone causes prostate cancer.29 A variety of testosterone products are on the market and a wide range of doses are recommended; however, the goal of replacement therapy should be just that, to replace the hormone that is missing. A healthy young man in his prime makes approximately 7 mg of testosterone/day.30 Using this as a guideline, physiologic doses of 7 to 10 mg/day of topical or sublingual testosterone are typically sufficient to affect metabolic processes in males with measured low or suboptimal levels.
In addition to direct replacement of testosterone, a number of natural therapies and concomitant treatment regimens can help to boost testosterone levels. As mentioned, the avoidance of endocrine-disruptors, including phthalates, BPA, PVC, and many compounds found in pesticides and fungicides, is very important for men of all ages. Agents that block the aromatase enzyme, and therefore reduce the conversion of testosterone into estrogen, can be a very effective intervention. Natural aromatase inhibitors include the bioflavonoids chrysin and luteolin, although it is important to note that studies appear to be more favorable for topical, rather than oral, chrysin.31 Zinc deficiency causes an upregulation of the aromatase enzyme; thus zinc supplementation will often blunt aromatase activity.32 Stinging nettle (Urtica dioica), an herb commonly used for allergies, can also be employed to bind to SHBG, which leaves more testosterone available to tissues.33 Finally, there are several herbs that can work to boost testosterone levels, including longjack (Eurycoma longifolia), horny goat weed (Epimedium grandiflorum), and tribulus (Tribulus terrestris).
Whether through limiting factors that adversely affect testosterone levels, natural therapies that help to boost testosterone production, or bioidentical hormone replacement, restoring hormone levels will both treat existing metabolic syndrome and also reduce the risk of developing it in the future. Comprehensive screening with a free serum or salivary testosterone level should be a part of standard work-up for any patient with symptoms consistent with metabolic syndrome.

Notes
1. Ervin RB. Prevalence of metabolic syndrome among adults 20 years of age and over, by sex, age, race and ethnicity, and body mass index: United States, 2003-2006. National Health Statistics Reports. No 13. Hyattsville, MD: National Center for Health Statistics; 2009.
2. Malik S, Wong ND, Franklin SS, et al. Impact of the metabolic syndrome on mortality from coronary heart disease, cardiovascular disease, and all causes in United States adults. Circulation. 2004. 110:1245-1250.
3. Corona G, Forti G, Maggi M. Why can patients with erectile dysfunction be considered lucky? The association with testosterone deficiency and metabolic syndrome. Aging Male. 2008 Dec; 11(4) 193-199.
4. Guay AT, Traish A. Testosterone deficiency and risk factors in the metabolic syndrome: implications for erectile dysfunction. Urol Clin North Am. 2011 May;38(2);175-183.
5. Miner MM. Men's health in primary care: an emerging paradigm of sexual function and cardiometabolic risk. Urol Clin North Am. 2012 Feb;39(1):1-23.
6. Mulligan T, Frick MF, Zuraw QC, Stemhagen A, McWhirter C. Prevalence of hypogonadism in males aged at least 45 years: the HIM study. Int J Clin Prac. 2006:60:762-769.
7. Zava D. Saliva hormone testing. Townsend Lett. 2004;246:120-124.
8. Vining RF, Mcinley RA. The measurement of hormones in saliva: possibilities and pitfalls. J Steroid Biochem. 1987 (27):81-94.
9. Dunn JF, Nisula BC, Rodbard D. Transport of steroid hormones: binding of 21 endogenous steriods to both testosterone-binding globulin and corticosteroid-binding globulin in human plasma. J Clin Endocrinol Metab. 1981;53:58-68.
10.Feldman HA, Longcope C, Derby CA, et al. Age trends in the level of serum testosterone and other hormones in middle-aged men: longitudinal results from the Massachusetts male aging study. J Clin Endocrinol Metab. 2002;87(2):589-598.
11.Cumming DC, Quigley ME, Yen SS. Acute suppression of circulating testosterone levels by cortisol in men. J Clin Endocrinol Metab. 1983 Sep;57(3):671-673.
12.Cicero TJ. Alcohol-induced deficits in the hypothalamic-pituitary-luteinizing hormone axis in the male. Alcohol Clin Exp Res. 1982;6:207-215.
13.Ye L, SU ZJ, Ge RS. Inhibitors of testosterone biosynthetic and metabolic activation enzymes. Molecules. 2011 Dec 2;16(12):9983-10001.
14.Basaria S. Androgen deprivation therapy, insulin resistance and cardiovascular mortality: an inconvenient truth. J Androl. 2008;29:534-539.
15.Traish AM, Saad F, Feeley RJ, Guay A. The dark side of testosterone deficiency: III. Cardiovascular disease. J Androl. 2009. (30)477-494.
16.Isidori AM, Caprio M, Strollo F, et al. Leptin and androgens in male obesity: Evidence for leptin contribution to reduced androgen levels. J Clin Endocrinol Metab. 1999;84;3673-3680.
17.Lebovitz HE, Banerji MA. Point: visceral adiposity is causally related to insulin resistance. Diabetes Care. 2005;28;9:2322-2325.
18.Rebuffe-Scrive M, Marin P, Bjorntorp P. Effect of testosterone on abdominal adipose tissue in men. Int J Obes. 1991 Nov;15(11):791-795.
19.Marin P, Oden B, Bjorntorp P. Assimilation and mobilization of triglycerides in subcutaneous abdominal and femoral adipose tissue in vivo in men: effects of androgens. J Clinical Endocrinol Metab 1995;80:239-243.
20.Kenny AM, Kleppinger A, Annis K, et al. Effects of transdermal testosterone on bone and muscle in older men with low bioavailable testosterone levels, low bone mass, and physical frailty. J Am Geriatr Soc. 2010 Jun;58(6):1134-1143.
21.Holmang A, Bjorntorp P. The effects of testosterone on insulin sensitivity in male rats. Acta Physiol Scand. 1992;146:505-510.
22.Malkin CJ, Pugh PJ, Kapoor D, Jones RD, Channer KS, Jones TH. The effect of testosterone replacement on endogenous inflammatory cytokines and lipid profiles in hypogonadal men. J Clin Endocrinol Metab. 2004;89:3313-3318.
23.Gorbachinsky I, Akpinar H, Assimos DG. Metabolic syndrome and urologic disease. Rev Urol. 2011;12(4):e157-e180.
24.Boyanov MA, Boneva Z, Christov VG. Testosterone supplementation in men with type 2 diabetes, visceral obesity and partial androgen deficiency. Aging Male. 2003;6:1-7.
25.Marin P, Holmang S, Jonsson L, et al. The effects of testosterone treatment on body composition and metabolism in middle-aged obese men. Int J Obes Relat Metab Disord. 1992;16:991-997.
26.Kalinchenko SY, Tishova YA, Mskhalaya GJ, Gooren LJ, Giltay EJ, Saad F. Effects of testosterone supplementation on markers of the metabolic syndrome and inflammation in hypogonadal men with the metabolic syndrome: the double-blinded placebo-controlled Moscow study. Clin Endocrinol (Oxf). 2010 Nov;73(5):602-612.
27.Tibblin G. Alderberth A. Lindstedt G. Bjorntorp P. The pituitary-gonadal axis and health in elderly men: a study of men born in 1913. Diabetes. 1996 Nov;45(11):1605-1609.
28.Kapoor D, Goodwin E, Channer KS, Jones TH. Testosterone replacement therapy improves insulin resistance, glycaemic control, visceral adiposity and hypercholesterolaemia in hypogonadal men with type 2 diabetes. Eur J Endocrinol 2006 (154);899-906.
29.Dobs AS, Morgentaler A. Does testosterone therapy increase the risk of prostate cancer? Endocr Pract. 2008 Oct;14(7):904-911.
30.Dandona P, Rosenberg MT. A practical guide to male hypogonadism in the primary care setting. Int J Clin Pract. 2010 May;64(6):682-696.
31.Kellis JT Jr, Vickery LE. Inhibition of human estrogen synthetase (aromatase) by flavones. Science. Sep 7 1984;225(4666):1032-1034.
32.Okubu T, Truong TK, Yu B, et al. Down-regulation of premotor I.3 activity of the human aromatase gene in breast tissue by zinc-finger protein, snail (SnaH). Cancer Res. 2001;61:1338.
33.Schottner M, Gansser D, Spiteller G. Lignans from the roots of Urtica dioica and their metabolites bind on human sex hormone binding globulin (SHBG). Planta Med. 1997 Dec;63(6):529-532.
34.Cirigliano MD, Szapary PO. Horny goat weed for erectile dysfunction. Alt Med Alert. 2001;4:19-22.
35.Prasad RNV. Sexual effects of puncturevine (Tribulus terrestris) extract (Protodioscin):An evaluation using a rat model. J Altern Comp Med. 2003 April 1;9(2):257-265.
36.Gauthaman K, Ganesan AP. The hormonal effects of Tribulus terrestris and its role in the management of male erectile dysfunction - an evaluation using primates, rabbit and rat. Phytomedicine. 2008;15(1-2):44-54.

 Dr. Wood grew up in Colorado and obtained her undergraduate degree in biochemistry from Colorado College. An enthusiasm for science but a passion for people led her to medicine, and a desire to treat the cause of disease, not just the symptoms, led her to naturopathy. After completing her doctorate at the National College of Naturopathic Medicine, Dr. Wood stayed in Oregon and has a private practice focused on endocrine imbalance, digestive dysfunction, immune support, and cardiovascular health. Dr. Wood grew up in Colorado and obtained her undergraduate degree in biochemistry from Colorado College. An enthusiasm for science but a passion for people led her to medicine, and a desire to treat the cause of disease, not just the symptoms, led her to naturopathy. After completing her doctorate at the National College of Naturopathic Medicine, Dr. Wood stayed in Oregon and has a private practice focused on endocrine imbalance, digestive dysfunction, immune support, and cardiovascular health.
In addition to her clinical practice, Dr. Wood is a staff physician with Labrix Clinical Services Inc., where she educates physicians and health care providers around the country about hormonal balancing through development of educational materials, contributions to a webinar series, and lectures at local and national conferences. In 2008 she coauthored a book on andropause titled His Change of Life: Male Menopause and Healthy Aging with Testosterone.
|



![]()
![]()
![]()



 Dr. Wood grew up in Colorado and obtained her undergraduate degree in biochemistry from Colorado College. An enthusiasm for science but a passion for people led her to medicine, and a desire to treat the cause of disease, not just the symptoms, led her to naturopathy. After completing her doctorate at the National College of Naturopathic Medicine, Dr. Wood stayed in Oregon and has a private practice focused on endocrine imbalance, digestive dysfunction, immune support, and cardiovascular health.
Dr. Wood grew up in Colorado and obtained her undergraduate degree in biochemistry from Colorado College. An enthusiasm for science but a passion for people led her to medicine, and a desire to treat the cause of disease, not just the symptoms, led her to naturopathy. After completing her doctorate at the National College of Naturopathic Medicine, Dr. Wood stayed in Oregon and has a private practice focused on endocrine imbalance, digestive dysfunction, immune support, and cardiovascular health.