In the US, 75 cents out of every health-care dollar is spent on chronic disease.1 However, few would argue against the fact that our medical system is failing to sufficiently address the chronic disease epidemic. A paradigm shift in health care is needed. The shift is found in integrative, individualized medicine, which marries state-of-the art laboratory analysis with a treatment approach that emphasizes dietary and nutritional interventions.
The case of ulcerative colitis below is from the soon-to-be released Case Studies in Integrative and Functional Medicine, published by Metametrix Institute. DNA analysis of the individual's gastrointestinal microbiota along with food allergy testing provided individualized guidance for interventions. It is highly unlikely that all cases of ulcerative colitis will present with the identical mix of contributing factors. Thus, specialized laboratory analysis is required to take us away from "one-size-fits-all" population-based medicine to a truly individualized approach that addresses the underlying causes of illness.
Case History
EM was 29 when she presented with severely active ulcerative colitis (UC). Colonoscopy had detected pancolitis (a severe form of UC that involves the whole colon. Pancolitis that is nonresponsive to medication frequently requires surgery).2 She was at her lowest body weight since entering adulthood. She was taking prednisone, mesalamine (a cyclooxygenase and prostaglandin blocker), and azathioprine (an immunosuppressive drug with unknown mechanism); but these medications were not effective. She had chronic diarrhea and severe fatigue. She also had erythema nodosum (nodular erythematous eruptions on the lower legs), which presented as large, multiple weeping lesions preceded by bruises. For the erythema nodosum, she was prescribed a two-month course of the antibiotic cephalexin. Because of the debility from her illness, EM stated that she couldn't mother her two children.
For most of her life, EM had followed the standard American diet (SAD) characterized by high intake of red meat, sugary desserts and drinks, high-fat foods, dairy products, eggs, and refined grains. She was previously evaluated for food allergies and determined to have reactions to yeast, gluten, and dairy, so she eliminated these components from her diet. Tests for celiac serology and genes turned out negative. Even with the adoption of this oligoantigenic diet based on her specific reactions, however, she noted no significant change in her condition at the time of her initial visit.
EM's family history did not include inflammatory bowel disease (IBD) or other autoimmune conditions. Her husband had irritable bowel syndrome. Although EM was under a great deal of stress because of her condition and had eaten the SAD for most of her life, there were no other contributing lifestyle factors apparent.
Laboratory Tests Ordered and Rationale
1. Previous testing: Tests for celiac serology and celiac genes turned out negative. Food allergy testing identified significant antibody elevations to yeast, wheat, and dairy. Adverse food reactions, including an association with celiac disease, have been identified in IBD.3,4 Removal of offending foods has been noted to improve symptoms.
2. Microbial stool analysis using DNA identification: Gastrointestinal microbiota imbalance (dysbiosis) is a common and significant finding in IBD.5 Dysbiosis was suspected to be a significant etiopathogenic factor in this case, particularly given the lack of favorable response to the removal of potential antigenic foods.
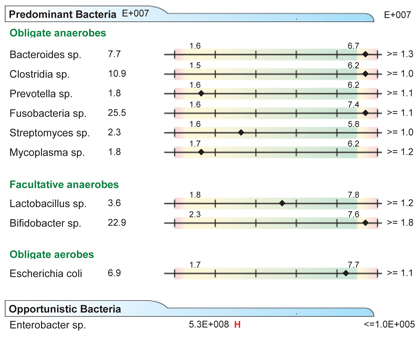
Figure 1: Microbial Stool Analysis Using DNA Identification.
Opportunistic bacteria present. Imbalanced predominant bacteria suggested dysbiosis.
Assessment
- Pancolitis with weight loss, fatigue, and diarrhea
- Erythema nodosum
- DysbiosisAntibiotic resistance
Treatment Plan
- Albendazole 250 mg: 1 tab p.o. b.i.d. for 3 days; then 1 tab on day 8, day 30, and day 90
- Berberine (botanical antimicrobial formula): 1 tab p.o. b.i.d.
- Multibotanical antimicrobial formula: 2 caps p.o. b.i.d. Contains: bilberry extract (25% anthocyanosides), noni, milk thistle, echinacea (purpurea and angustifolia), goldenseal, shiitake, white willow (bark), garlic, grape-seed extract, black walnut (hull and leaf), raspberry, fumitory, gentian, tea tree oil, galbanum oil, lavender oil (plant and flower), oregano oil
- Deglycyrrhizinated licorice (DGL): 2 tabs p.o. t.i.d. a.c.
- Aloe vera capsules: 2 caps p.o. b.i.d.
- Vegetable greens powder: 1 scoop p.o. q.d.
- Lactobacillus rhamnosus strain GG
- Saccharomyces boulardii
- Lactobacillus sp., Bifidobacter sp., and Streptococcus thermophilus
- Digestive enzymes: 2 tabs p.o. t.i.d. a.c.
Continue:
- Diet: Eliminate gluten, dairy, and yeast
- Mesalamine (COX/PG blocker) 400 mg: 1 tab p.o. t.i.d.
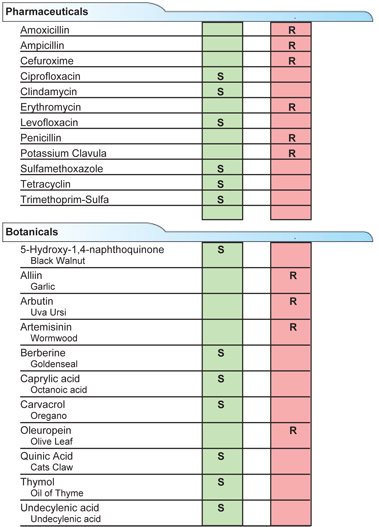
Treatment plan rationale: It was decided that hookworm (Necator americanus) would be treated with albendazole and the pathogenic and opportunistic bacteria with antimicrobial botanicals that were identified as sensitive upon laboratory testing (Figure 3). Botanicals were chosen because the patient had a history of extensive exposure to antibiotics. Botanicals may be less disruptive to the predominant flora for a number of reasons. For example, some antimicrobial botanicals contain prebiotic substances such as polysaccharides.6 Botanicals are also suggested to be less likely to contribute to antibiotic resistance.7 The mucilaginous botanicals aloe vera and licorice (DGL) were used to reduce GI inflammation and support healing of the mucosa.8,9 Comprehensive probiotics, including Saccharomyces boulardii, were used to normalize microflora, reduce inflammation, and support intestinal health.10The dietary restrictions and mesalamine were continued.
Ten-Month Follow-Up
EM gained weight and the erythema nodosum resolved. Her energy level increased. She had stopped the azathioprine and had been titrated off the steroids. Her dose of mesalamine was reduced and then discontinued, although she occasionally used it for flare-ups. The flare-ups were mostly caused by inadvertent exposure to foods she was reactive to. Although these exposures would throw her off track for a few days, she recovered more quickly than she had in the past. She thought that if she were successful in avoidance of all reactive foods, her recovery would be 100%. To EM, the most important benefit of her healing was that she could once again mother her two children. As she herself said, she was doing "incredibly well." For maintenance treatment, it was recommended that EM continue with dietary restrictions, probiotics, and digestive enzymes; periodic clinical and laboratory assessments would determine need for additional interventions.
Ten-Month Follow-up Laboratory Results (Not shown)
Findings were negative for Enterobacter sp., Campylobacter sp., EHEC, and hookworm. Saccharomyces species were detected, probably as a result of supplementation. A "taxonomy unavailable" parasite was detected, probably from dietary exposure to nonpathogenic parasites. Predominant bacteria were more balanced, although Bacteroides species was low.
Figure 3: Bacterial Sensitivities to Pharmaceutical and Botanical Interventions.
Findings suggested that bacteria detected in stool may have resistance to cefuroxime, among other antibiotics and botanicals. S = Sensitive; R = Resistant.
Discussion
EM's pancolitis and erythema nodosum were evidence of an intense systemic inflammatory reaction. Factors involved in the pathogenesis of IBD include the host's intestinal microflora, the host's genetic and immune predisposition, infection, psychological stress, environmental stimuli, and intestinal barrier permeability. 11-13 Immunological reactivity to foods, including gluten intolerance, have been reported as causes of colonic inflammation and IBD.3,4
EM did not have a family history of IBD, but she was under a good deal of stress caused by her limited ability to care for her children. While this particular stressor was not a causative factor, it may have exacerbated her IBD. Components of the SAD she followed for most of her life were very antigenic for her, as identified on food allergy testing. The severe reactions reflected a pronounced inflammatory response. This may have initiated immune responses similar to those triggered by IgG4 antibodies. IgG4 is an immunoglobulin that forms a complex with the offending food protein. Antigen–antibody complexes may initiate an immune response, leading to a wide variety of symptoms, ranging from gastrointestinal symptoms including diarrhea to skin changes, headaches, or joint inflammation and myopathy. Indeed, symptoms associated with IgG reactions are found associated with most organ systems.14
This patient's laboratory results pointed to several factors that may have contributed to the pathogenesis of UC. As shown in Figure 1, very high levels of predominant bacteria spp. (Bacteroides sp., Clostridia sp., Fusobacteria sp., and Bifidobacter sp.) and Enterobacter sp. (an opportunistic bacterium) demonstrated significantly altered microflora (dysbiosis). Imbalances of predominant anaerobic bacteria (both high and low) have been reported in IBD. Specifically, Bacteroides, Eubacterium (such as E. coli, Enterobacter, Shigella, Salmonella, Yersinia, etc.), and Lactobacillus have been identified.15 Similar to EM's findings, studies have also shown increased anaerobes (especially gram-negative anaerobes such as Bacteroides) in Crohn's disease (CD), UC, and pouchitis. In one study, 50% of CD patients had either E. coli or Bacteroides as the predominant GI bacterial group.16
Enterobacter sp., the opportunistic bacterium shown in Figure 1,is asymptomatic in healthy individuals but a leading cause of nosocomial bacterial infections, including urinary tract infections, bacteremia, skin and soft tissue infections, and septic arthritis.17 However, there are not many cases in the literature of Enterobacter-induced colitis or gastroenteritis.
Figure 2: Microbial Stool Analysis Using DNA Identification.
Pathogenic bacteria and a parasite detected in patient's stool.
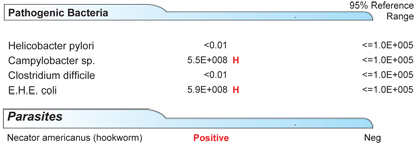
Two pathogens, Campylobacter sp. and Enterohemorrhagic Escherichia coli (EHEC), were also detected (Figure 2), illustrating the severity of dysbiosis. Bacterial enteritis can induce or exacerbate IBD.12 Campylobacter sp. and EHEC have been reported as causes of bacterial colitis.13,18,19 Campylobacter specifically has been detected in IBD and has been cited as a rare cause of pancolitis.18,20
Erythema nodosum is the most common dermatologic manifestation of IBD, and UC specifically is known to trigger this condition, especially during flare-ups.21 Further, erythema nodosum can be caused by bacterial or fungal infection, and Campylobacter sp. has been specifically implicated.21 Erythema nodosum has also been associated with celiac disease.22 In this case, it may be that the combination of immunosuppressive steroids, IBD, GI Campylobacter,and gluten sensitivity led to the expression of this condition.
Hookworm was detected (Figure 2). UC has been exacerbated or even caused by helminth infections, specifically Strongyloides species.23,24 However, "worm therapy" using the helminth Trichuris suis has been reported to significantly improve IBD symptoms.25
Based on research findings, Campylobacter sp. and EHEC were most likely involved in the pathogenesis of EM's pancolitis. Given the dearth of evidence connecting Enterobacter sp. and Necator americanus to pancolitis, it is not clear the contribution that these made in EM's condition, although they may have added to her overall inflammatory burden.
As EM's remarkable clinical response and normalized follow-up laboratory results demonstrate, she responded favorably to treatment. Consistent with published research, the aggressive protocol of probiotics appears to have been quite therapeutic in EM's case.15,16,26 Treatments with Lactobacillus sp., Bifidobacteria sp., and Saccharomyces boulardii have been shown to bring about remission in UC.15
It is interesting to note that the removal of antigenic foods from EM's diet appeared to have clearly contributed to her improvement, as evidenced by the fact that accidental ingestion of those substances triggered symptomatic flare-ups. However, it was only after normalization of bowel microbial status that the effect of antigenic foods was experienced, suggesting that the GI microbial imbalances in EM were the significant underlying etiological factors
Conclusion
A comprehensive protocol designed to remove GI microbial imbalances and antigenic foods, replace digestive enzymes and nutrients, repair the intestinal lining, and reinoculate with beneficial microflora resulted in almost a full recovery from the initial presentation. The patient experienced a significant improvement or resolution of all of her major symptoms, including diarrhea, erythema nodosum, fatigue, and weight loss; and she was able to stop regular use of all medications. She observed that it was likely that hidden exposure to antigenic foods resulted in the periodic flare-ups that kept her from a full, 100% recovery.
The fact that this UC case showed such dramatic improvement once the microbiota were normalized strongly suggests that the approach used in this case might have great benefit for any inflammatory bowel case, particularly in light of the numerous reports in the literature that significant microbial imbalances are associated with the etiopathogenesis of IBD. Given the epidemiological statistics of IBD, research investigating the efficacy of this cost-saving, safe, multifactorial, and integrative approach is highly recommended.

Notes
1. Center for Health Policy CUSoN. The Public Health Workforce: Enumeration 2002. Washington, DC: Department of Health and Human Services, Health Resources and Services Administration; 2000.
2. Hanauer SB. Inflammatory bowel disease: epidemiology, pathogenesis, and therapeutic opportunities. Inflamm Bowel Dis. Jan 2006;12 Suppl 1:S3–S9.
3. Yang A, Chen Y, Scherl E, Neugut AI, Bhagat G, Green PH. Inflammatory bowel disease in patients with celiac disease. Inflamm Bowel Dis. Jun 2005;11(6):528–532.
4. MacDermott RP. Treatment of irritable bowel syndrome in outpatients with inflammatory bowel disease using a food and beverage intolerance, food and beverage avoidance diet. Inflamm Bowel Dis. Jan 2007;13(1):91–96.
5. Marteau P. Bacterial flora in inflammatory bowel disease. Dig Dis. 2009;27 Suppl 1:99–103.
6. Guo FC, Williams BA, Kwakkel RP, et al. Effects of mushroom and herb polysaccharides, as alternatives for an antibiotic, on the cecal microbial ecosystem in broiler chickens. Poult Sci. Feb 2004;83(2):175–182.
7. Abascal K YE. Herbs and Drug Resistance: Part 1—Herbs and Microbial Resistance to Antibiotics. Alternative and Complementary Therapies. August 2002;8(4):237–241.
8. Davis RH, Donato JJ, Hartman GM, Haas RC. Anti-inflammatory and wound healing activity of a growth substance in Aloe vera. J Am Podiatr Med Assoc. Feb 1994;84(2):77–81.
9. Stewart PM, Prescott SM. Can licorice lick colon cancer? J Clin Invest. Apr 2009;119(4):760–763.
10. Mengheri E. Health, probiotics, and inflammation. J Clin Gastroenterol. Sep 2008;42 Suppl 3 Pt 2:S177–178.
11. Goodhand JR, Wahed M, Rampton DS. Management of stress in inflammatory bowel disease: a therapeutic option? Expert Rev Gastroenterol Hepatol. Dec 2009;3(6):661–679.
12. Kalischuk LD, Buret AG. A role for Campylobacter jejuni-induced enteritis in inflammatory bowel disease? Am J Physiol Gastrointest Liver Physiol. Oct 29 2009.
13. Singh J. Colitis [Web article]. WebMD. http://emedicine.medscape.com/article/927845-overview. Accessed 12/31/09, 2009.
14. Lord RS BJ. Laboratory Evaluations for Integrative and Functional Medicine. Atlanta: Metametrix Institute; 2008:435–437.
15. Ott SJ, Musfeldt M, Wenderoth DF, et al. Reduction in diversity of the colonic mucosa associated bacterial microflora in patients with active inflammatory bowel disease. Gut. May 2004;53(5):685–693.
16. Tamboli CP, Neut C, Desreumaux P, Colombel JF. Dysbiosis in inflammatory bowel disease. Gut. Jan 2004;53(1):1–4.
17. Fraser SL, Arnett M. Enterobacter infections [Web article]. WebMD http://emedicine.medscape.com/article/216845-overview. Accessed Jan. 5, 2010.
18. Siegal D, Syed F, Hamid N, Cunha BA. Campylobacter jejuni pancolitis mimicking idiopathic ulcerative colitis. Heart Lung. Jul–Aug 2005;34(4):288–290.
19. Sumrall A, McMullan L, Abrasley C, East H. A case of severe hemorrhagic diarrhea. J Miss State Med Assoc. Dec 2007;48(12):366–369.
20. Gradel KO, Nielsen HL, Schonheyder HC, Ejlertsen T, Kristensen B, Nielsen H. Increased short- and long-term risk of inflammatory bowel disease after salmonella or campylobacter gastroenteritis. Gastroenterology. Aug 2009;137(2):495–501.
21. Hebel JL, Habif T. Erythema nodosum [Web article]. WebMD. http://emedicine.medscape.com/article/1081633-overview. Accessed Dec. 31, 2009.
22. Bartyik K, Varkonyi A, Kirschner A, Endreffy E, Turi S, Karg E. Erythema nodosum in association with celiac disease. Pediatr Dermatol. May–Jun 2004;21(3):227–230.
23. Ghoshal UC, Alexender G, Ghoshal U, Tripathi S, Krishnani N. Strongyloides stercoralis infestation in a patient with severe ulcerative colitis. Indian J Med Sci. Mar 2006;60(3):106–110.
24. Weight SC, Barrie WW. Colonic Strongyloides stercoralis infection masquerading as ulcerative colitis. J R Coll Surg Edinb. Jun 1997;42(3):202–203.
25. Summers RW, Elliott DE, Urban JF, Jr., Thompson RA, Weinstock JV. Trichuris suis therapy for active ulcerative colitis: a randomized controlled trial. Gastroenterology. Apr 2005;128(4):825–832.
26. Sheil B, Shanahan F, O'Mahony L. Probiotic effects on inflammatory bowel disease. J Nutr. Mar 2007;137(3 Suppl 2):819S–824S.

 Kara N. Fitzgerald, ND, graduated from NCNM [National College of Natural Medicine] and completed postdoctorate and residency training at Metametrix Clinical Laboratory and Progressive Medical Centers in Atlanta, Georgia. She is author and editor of Case Studies in Integrative and Functional Medicine and contributing author to Laboratory Evaluations in Integrative and Functional Medicine. She also serves on the faculty of Institute for Functional Medicine and maintains a private practice at Advanced Diagnostic, in New Haven, Connecticut. Kara N. Fitzgerald, ND, graduated from NCNM [National College of Natural Medicine] and completed postdoctorate and residency training at Metametrix Clinical Laboratory and Progressive Medical Centers in Atlanta, Georgia. She is author and editor of Case Studies in Integrative and Functional Medicine and contributing author to Laboratory Evaluations in Integrative and Functional Medicine. She also serves on the faculty of Institute for Functional Medicine and maintains a private practice at Advanced Diagnostic, in New Haven, Connecticut.
 Cass Nelson-Dooley, MS, is the research coordinator at the Metametrix Institute and educates clinicians about the use of laboratory data in patient care. Ms. Nelson-Dooley completed a Fulbright Scholarship and master of science in ethnopharmacology. She is coauthor of "Nutrient and Toxic Elements" in Laboratory Evaluations for Integrative and Functional Medicine and has published articles in the Journal of Nutrition, Obesity, and Current Medicinal Chemistry. Cass Nelson-Dooley, MS, is the research coordinator at the Metametrix Institute and educates clinicians about the use of laboratory data in patient care. Ms. Nelson-Dooley completed a Fulbright Scholarship and master of science in ethnopharmacology. She is coauthor of "Nutrient and Toxic Elements" in Laboratory Evaluations for Integrative and Functional Medicine and has published articles in the Journal of Nutrition, Obesity, and Current Medicinal Chemistry.
Kathleen Janel, ND, contributed the above case to Case Studies in Integrative and Functional Medicine. She practices specialty natural medicine with a focus on digestive medicine in Mukilteo, Washington. She states that her practice philosophy is to bridge the gap between natural and conventional medicine, using both natural and pharmaceutical interventions, depending on need. Learn more at gastricmedicine.com.
|



![]()
![]()
![]()




 Kara N. Fitzgerald, ND, graduated from NCNM [National College of Natural Medicine] and completed postdoctorate and residency training at Metametrix Clinical Laboratory and Progressive Medical Centers in Atlanta, Georgia. She is author and editor of
Kara N. Fitzgerald, ND, graduated from NCNM [National College of Natural Medicine] and completed postdoctorate and residency training at Metametrix Clinical Laboratory and Progressive Medical Centers in Atlanta, Georgia. She is author and editor of  Cass Nelson-Dooley, MS, is the research coordinator at the Metametrix Institute and educates clinicians about the use of laboratory data in patient care. Ms. Nelson-Dooley completed a Fulbright Scholarship and master of science in ethnopharmacology. She is coauthor of "Nutrient and Toxic Elements" in
Cass Nelson-Dooley, MS, is the research coordinator at the Metametrix Institute and educates clinicians about the use of laboratory data in patient care. Ms. Nelson-Dooley completed a Fulbright Scholarship and master of science in ethnopharmacology. She is coauthor of "Nutrient and Toxic Elements" in 