Online publication only
Introduction
Tom sat in his dormitory room for hours and hours. Soon the sun went down and the room became dark and gloomy. His depression became despair as he sank lower and lower, despite the antidepressants he'd been taking for years.
After mustering enough energy, Tom lifted himself off the seat and wandered over to the small refrigerator. He found some cheese and an energy drink. He sat down at his desk and ate his meager meal. Then the fatigue drove him to the bed, where he lay until morning.
The next day Tom awoke with even more fatigue. He immediately walked over to the refrigerator and guzzled two more energy drinks. The raging hammers in his head were driving him to do something violent. He went over to the nightstand and pulled out a handgun and a box of ammunition. Then Tom went out of his dormitory room, entered the first open door he came to, and opened fire.
Was Tom evil? Was he crazy or sick? Too bad he didn't know about Whole Body Biometabolic Analysis and the "balanced brain." Perhaps he could have been helped and many people's lives would have been saved.
Before we can understand the balanced brain, we need to understand how the brain works. The nervous system is like a network of telephone wires that connect conversations between different parts of the body to each other and to the brain. Connecting telephone wires to different houses is done by relays. Similarly, in the nervous system, chemical messengers pass the signals from one nerve to another. These chemical messengers are called neurotransmitters. The two primary neurotransmitters of interest are serotonin and norepinephrine.
What happens when they are not in balance? When serotonin is low, a number of disorders will occur. When serotonin and norepinephrine are low, one will experience depression. When serotonin and norepinephrine are very low, one will experience a migraine headache. When serotonin is low and norepinephrine is high, one will experience increased agitation and perhaps violence. Chemical stimulants such as caffeine, ephedrine, and pseudoephedrine can produce an increase in norepinephrine, which can result in mania, exaggerated aggression, and perhaps violence if serotonin in the brain is low.
Tyramine, the amino acid contained in fermented products such as cheese, when ingested causes the loss of serotonin and norepinephrine.
This is just the tip of the iceberg of the Biometabolic Analysis system.
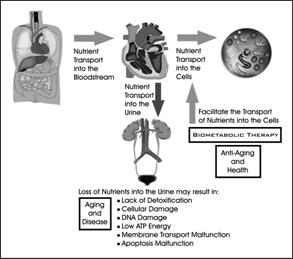
Many practitioners focus on a single aspect an individual's health while ignoring other parts of the body. Often a treatment plan deals with the symptoms of a single problem and not the original cause or causes. The human body is not simple enough for a single problem to exist by itself without affecting other parts of the body. Biometabolic Analysis is a scientific system that addresses aspects of the entire body and their interactions. Similarly, nutritionists normally deal with the transport of nutrients from the intestines into the bloodstream. Getting the nutrients from the bloodstream into the cells is assumed to function by itself without problems.
People may be eating right and getting plenty of exercise, but if the critical process of transporting nutrients into the cells is not taking place, they're just making expensive urine! If nutrients fail to get into the cells, the lactic acid buildup within the cells can damage the cell membrane and DNA, which is the reason that the body ages, organs degrade, and diseases begin.
What Causes These Pathways to Change?
Damage to the cells from toxins is the major cause of many health problems. Detoxifying and ridding the body of toxins, particularly neurotoxins, which deplete serotonin and norepinephrine, is an important part of balancing serotonin and norepinephrine levels.
It is very important to avoid toxic chemicals from the environment. Tap water should be filtered to remove lead, chlorine, heavy metals, and bacteria. It is also important to consume an adequate supply of antioxidants to prevention cellular damage.
Toxicity from foreign chemicals (exotoxins) can damage almost all cells of the body. Symptoms include fatigue, headaches, neurological disorders, chemical sensitivities, immune dysfunction, and liver disorders. Food is often the main source of toxins. There are thousands of chemicals used by the food industry during processing and packaging. Many farmers use pesticides, which are passed to consumers. In addition to these external sources of toxins, the body also produces toxins internally called endotoxins, resulting from digestion, immune system functions, emotional stress, and so on. Endotoxins may also be produced as the result of food allergies and sensitivities.
Fat-soluble toxins are easily absorbed but poorly excreted. Often, they accumulate in the body, damaging the tissues and organs, and depleting serotonin and norepinephrine. Fat-soluble chemicals are converted to water-soluble chemicals, primarily in the liver and in some cells, in a two-step process so that the water-soluble toxins can be excreted by the urine, liver, and skin. The skin is a vitally important organ that eliminates toxins through perspiration.
Detoxification Pathways
During the first phase of detoxification, fat-soluble chemicals are converted into intermediate chemicals. As a result of this process, free radicals are produced. The free radicals and the intermediate chemicals can damage the cells. An adequate quantity of antioxidants must be present to detoxify these intermediate compounds produced during the first phase of detoxification. The first phase may also detoxify some chemicals directly without requiring a second phase conversion.
During the second phase, the intermediate chemicals are converted into water-soluble chemicals, which are less toxic and easily excreted in the urine, bile, and skin.
The ability of the liver to detoxify is determined by the availability of the appropriate nutrients and enzymes. An adequate supply of antioxidants is vitally important after the first phase of converting fat-soluble toxins, which produce free radicals. Reduced glutathione, superoxide dismutase, and catalase are the primary antioxidants used in the body to neutralize free radicals. Other antioxidants include beta-carotene, vitamin E, vitamin C, selenium, N-acetylcysteine, lipoic acid, and proanthocyanidins. Vitamin and mineral cofactors required for cytochrome P450 reactions include riboflavin, niacin, magnesium, iron, and other trace minerals. Phytochemicals such as indoles from cruciferous vegetables and quercetin also help during the first phase of detoxification. Other second-phase conjugating agents include amino acids such as glycine, cysteine, glutamine, methionine, taurine, glutamic acid, and asparatic acid.
Vitamin, mineral, and protein deficiencies will decrease the activity of the detoxification pathways. Fats and polyunsaturated oils can promote the uptake of many chemical carcinogens in the gastrointestinal tract. Olive oil (monounsaturated) and omega-3 polyunsaturated oils (EPA, DHA) have a neutral effect in promoting the uptake of carcinogens in the gastrointestinal tract.
Figure 1
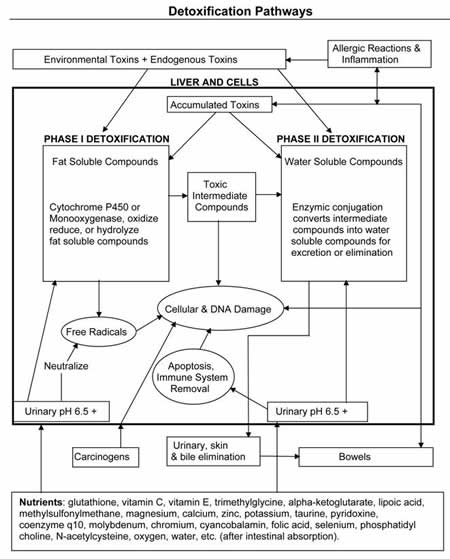
As previously discussed, the detoxification process requires various nutrients to function. Without such nutrients in the cells, intermediate compounds can cause cellular and DNA damage. If the apoptosis system (i.e., cells self-destruct if they are damaged) does not get the proper nutrients, the damaged cells can reproduce. If the immune system does not get the proper nutrients, the damaged cells can reproduce out of control, causing cancer.
Transporting the nutrients into the blood is only the first step. The nutrients must enter the cells in order to be available for use by the cells. For the nutrients to get into the cells, they must be transported though the cell membrane. When there is a lack of nutrients, particularly oxygen inside the cell, there is a buildup of lactic acid inside the cell. Excessive lactic acid damages the cell membrane transport mechanism and DNA. Lactic acid causes the cell to become acidic (lower pH). There is a correlation between intracellular pH and urine pH. The urine pH must be 6.5 or greater, which indicates the maximum amount of lactic acid in the cell for the membrane transport mechanisms to function at peak performance.
Tyramine Depletion of Serotonin and Norepinephrine
Tyramine displaces norepinephrine from sympathetic nerve endings and epinephrine from the adrenal glands. Initially, this release results in increased sympathetic stimulation, which causes higher blood pressure, insomnia, increased aggression in some cases, increased glucose release, increased metabolism, inhibition of the gastrointestinal tract, and increased cardiovascular activity. Increased sympathetic stimulation can also result in a depletion of serotonin. The final result is a depletion of norepinephrine, epinephrine, and serotonin reserves. An individual's activity, as a result of tyramine intake, may initially go from mania to depression and migraine headaches later on. The presence of monoamine oxidase in the gastrointestinal tract will inactivate tyramine. Compounds with tyraminase properties will be helpful in the treatment of lost serotonin and norepinephrine after tyramine ingestion. So far, none have been found.
The tyramine content in foods varies greatly due to different processing, aging, fermentation, ripening, and/or contamination. Many foods that originally contained small amounts of tyramine develop large amounts if left to spoil, age, or ferment. For example, bananas are permissible if they are fresh, not if they are overripe. Fruits that are permissible should be very fresh. Avoid leftovers kept in the refrigerator, especially meats; dry packaged mixes; canned products; yeast extracts; and protein extracts. Tyramine can also be produced by bacteria in the gastrointestinal tract, including Helicobacter pylori and even probiotics in the gut.
Mild tyramine reactions can occur almost immediately after the ingestion of approximately 6 mg of tyramine in a single portion. Severe reactions can occur almost immediately after the ingestion 10 to 25 mg of tyramine. Blood pressure will begin to rise within several hours after ingestion of tyramine.
Approximate tyramine contents of food:
- Cheddar cheese: 1.5 mg/g (42.5 mg/ounce)
- Blue Stilton: 0.2 mg/g (5.6 mg/ounce)
- Gouda: 0.02 mg/g (0.56 mg/ounce)
- Beer: 0.02 mg/g (0.56 mg/ounce)
- Wine: 0.025 mg/g (0.71 mg/ounce)
- Yeast extracts: 2 mg/g (56.6 mg/ounce)
The Process of Aging and Disease
Figure 2
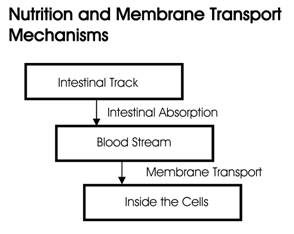
Figure 3
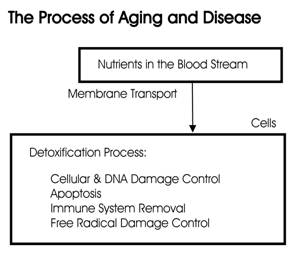
The reproduction of damaged cells and DNA is the reason that the body ages, organs degrade, and diseases begin. Accumulated toxins and excessive lactic acid inside the cells damage the cells and DNA when detoxification systems do not adequately function.
Nontoxic food can also affect body chemistry and produce toxins. Foods with a high glycemic index such as sugar and starch will stimulate the body to produce too much insulin and cholesterol, which leads to inflammation, constricted blood vessels, pain, acidosis, and disease.
Acidosis is a condition whereby there is too much lactic acid in the cells, which eventually causes cellular, DNA, and membrane transport system damage. If the cells' DNA is damaged, apoptosis is not functioning properly, and the immune system does not remove the cell, it will reproduce. The continued reproduction of DNA-damaged cells becomes a cancerous tumor.
Biochemical and Biophysics Pathways of Cellular Metabolism
Figure 4

Oxygen deficiency in cells results primarily from inadequate mechanism of transport through the cell membrane, rather than the amount of oxygen in the blood, except in cases where athletic activity does not supply enough oxygen to the blood. Energy is produced through the glycolytic pathway. In glycolysis, glucose is converted to glucose-6-phosphate, then to glycogen and/or pyruvate and 2 molecules of ATP (energy). Glycolysis does not require any oxygen (anaerobic). In the absence of oxygen, the process of oxidation and phosphoryation consumes the cells' supply of NAD+ (nicotinamide adenine dinucleotide). NADH can be reoxidized by converting pyruvate to lactate in the presence of lactic dehydrogenase (LDH), which favors the conversion of pyruvate to lactate. In the presence of oxygen, pyruvate undergoes oxidative decarboxylation whereby pyruvate + NAD+ + CoA is converted to acetyl CoA + NADH + CO2. Acetyl CoA is the beginning of the Krebs (citric acid) cycle, which produces 38 molecules of ATP (energy), carbon dioxide, and water, through oxidative phospholoryation (aerobic). The five coenzymes needed for these reactions are thiamin pyrophosphate, lipoic acid, CoA, FAD, and NAD+. The four water-soluble vitamins needed are thiamin, riboflavin, nicotinic acid, and pantothenic acid. If there is a lack of oxygen in the cell, there will be a buildup of lactic acid, which can dramatically alter the pH of the cell to be more acidic. In the acidic environment, enzymes from lysosomes within the cell are released and react with the acidic surroundings, which may prevent the membrane double bonds from reaching higher excitation levels and eventually damage the cells' DNA.
Figure 5
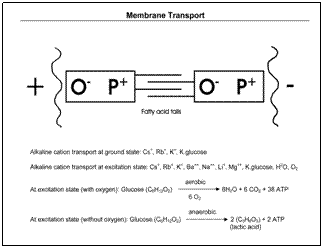
The rate of glucose transport into the cells increases to as much as 10 times in the presence of insulin. Excessive amounts of insulin, which can occur when consuming high glycemic index foods, can create excessive amounts of glucose in the cells. Without adequate oxygen in the cells, there can be a buildup of lactic acid, which can dramatically alter the pH of the cell to be more acidic. In the acidic environment, enzymes from lysosomes within the cell are released and react with the acidic surroundings, which may prevent the double bonds from reaching higher excitation levels and eventually damage the cells' DNA.
The most important part of the cell membrane is the phosphorous and oxygen double bond (P=O). Four electrons are closer to the oxygen atom than the phosphorous atom, which creates an electrical potential (P+=O−). This electrical potential can cause cations to enter or leave a cell. The cell membrane contains a double layer of these bonds, connected by fatty acid tails, creating complex pumping action in and out of the cells. The strength of the attraction depends upon the excitation level of double bond in the cell membrane. At the lowest excitation level (ground state) of the double bond, it produces the smallest electrical potential across the cell membrane. At the ground state, the cell membrane can only transport cesium+, rubidium+, and potassium+ alkaline cations into the cell. Therefore, the membrane can transport primarily water and glucose. In the ground state, the cell membrane will have great difficulty in transporting oxygen into the cells. As previously described, there will be a buildup of lactic acid, which changes the cellular pH. The energy required to increase the excitation level of the double bond is produced within the cell. At the higher energy levels, the four electrons of the double bond are pulled farther from the phosphorus atom, which results in higher electrical potentials across the cell membrane, which results in greater transport of molecules into the cell. At the high excitation levels of the double bond, the cell membrane can transport water, glucose, oxygen, and other nutrients into the cell.
Carcinogens are chemicals with certain electrical properties that coat the cell membrane, which prevent the higher excitation levels of the double bonds, which results in mostly water and glucose being transported into cells. The cells become deficient in oxygen and other nutrients. The cancer process begins with the loss of approximately one-third of the oxygen transport into the cell. Anaerobic metabolism causes a buildup of lactic acid within the cell, which dramatically changes the pH of the cell to acidic. In the acidic environment, enzymes from lysosomes within the cell are released and react with the acidic surroundings, which may prevent the double bonds from reaching higher excitation levels. The acidic environment will eventually damage the DNA within the cells resulting in a loss of the usual genetic controls whereby cellular size and shape are no longer regulated.
The normal pH range for cells is about 7.3 to 7.4. Death occurs when the blood pH falls to 7.0 or rises to 8.0. Intracellular pH ranges from 6.0 to 7.4. If the cell drops below 6.0 or rises above 8.0, the cell dies. Normal cells can maintain this narrow range of pH through the pH buffering system, which consists of sodium bicarbonate (NaHCO3) and carbonic acid (pCO3) in the same solution. In the intracellular fluid, there is little sodium bicarbonate, and the bicarbonate ion occurs primarily as potassium and magnesium bicarbonate. There are also phosphate and protein buffering systems. In the low excitation state, the double bonds may not be able to transport enough molecules to significantly change the pH of a very acidic cellular environment.
What is Biometabolic Analysis?
This revolutionary system analyzes biochemical and biophysics pathways and facilitates the transport of nutrients into the cells, not just the absorption from the intestines into the blood stream. The program will produce a step-by-step plan geared towards each person's needs to facilitate the appropriate changes for the problems discovered, including migraine, depression, natural hormone replacement, and multiple disorders developing over time, which affect the total body chemistry and biophysics of the individual. The biochemical and biophysical pathways, which change over time, are addressed with the Biometabolic Analysis.
This is a unique 22,000-line algorithm software designed to statistically correlate the nutritional protocols to the patient's diagnostic data collected, with the objective of balancing the biochemical and biophysics pathways leading to the following conditions:
Membrane Transport Mechanisms
Hypoimmune (low)
Respiratory allergies
Food allergies
Heart
Circulation
Blood pressure
Asthma
Stomach/gastritis/ulcer
Small intestine/pancreas
Hypoglycemia (low glucose)
Diabetes
Intestinal candida overgrowth
Colon
Irritable bowel
Increased intestinal permeability
Liver/gall bladder
Malnutrition
Actual weight (pounds)
Maximum reference weight (pounds)
Hypothryroid (underactive)
Hyperthryroid (overactive)
Hypoadrenal (underactive)
Hyperadrenal (overactive)
Rheumatoid arthritis
Osteoporosis
Peripheral neuropathy
Migraine
Brain function abnormalities
Kidney/bladder
Hidden microorganisms
Men: prostate, male reproduction, testosterone lack, testosterone excess
Women: premenstrual syndrome, dysmenorrhea, menopause, estrogen excess, estrogen lack, progesterone lack
Retinol (vitamin A) deficiency
Calciferol (vitamin D) deficiency
Tocopherol (vitamin E) deficiency
Thiamine (vitamin B1) Deficiency
Riboflavin (vitamin B2) deficiency
Niacin (vitamin B3) deficiency
Pyridoxine (vitamin B6) deficiency
Cobalamin (B12 def/pernicious anemia)
Pantothenic acid deficiency
Biotin deficiency
Folic acid deficiency
Ascorbic acid (vitamin C) deficiency
Calcium deficiency
Phosphorus deficiency
Magnesium deficiency
Potassium deficiency
Iron deficiency (microcytic anemia)
Copper deficiency
Iodine deficiency
Zinc deficiency
Protein deficiency
Essential fatty acid deficiency
Hypochlorhydria (low stomach acid)
Depression
Mania
Obsessive-compulsive disorder
Figure 6: Transport of Nutrients
The data collected that provides the highest level of correlation would include:
Symptoms
Height and weight
Zinc taste test
Blood pressure
Daytime core temperature
Urinalysis
Salvia pH
Bioelectric impedance analysis
Blood chemistry
Nutritional microscopy
Conclusion
The results of clinical investigation led to substantial improvement of health where the recommendations of the Biometabolic Analysis were followed. For further information and sample reports, see my website: www.allocca.com

Dr. John A. Allocca is a medical research scientist. His most current work includes the textbook Topics in Clinical Nutrition, a computerized biochemical analysis and nutritional plan software program, and nutritional formulas for migraine and other disorders. His previous work includes a computerized neonatal diagnostic system, a method for measuring intracranial pressure and vascular compliance noninvasively, development of computerized medical diagnostic systems and rehabilitative instruments. He received a doctor of philosophy degree in clinical nutrition from St. Martin University College of Health Science in 1997, a doctor of science degree in biophysics from Pacific Western University in 1981, a master of science degree in bioengineering from Polytechnic Institute of New York in 1979, and completed coursework and dissertation for a doctor of philosophy in physics at Columbia Pacific University in 1984. He has published numerous textbooks in medicine, nutrition, physics, and electronics. He has also published numerous papers in medicine, nutrition, and physics.
Dr. Allocca is a full honored member of Sigma Xi: The Research Society, the American Society for Microbiology, IEEE, Engineering in Medicine and Biology Society, and New York Academy of Sciences. He has been practicing nutrition since 1984. He became a certified clinical nutritionist (CCN) in 1996. His biography can be found in Who's Who in Frontier Science and Technology and Who's Who in the World.
19 Lorraine Court
Northport, NY 11768
631-757-3919
john@allocca.com
www.allocca.com

Bibliography
Allocca JA. Balancing Serotonin and Norepinephrine Levels: The Treatment of Choice. Allocca Biotechnology LLC; 2007.
———. The Migraine-Depression Solution. Allocca Biotechnology; 2006.
———. Topics in Clinical Nutrition. Allocca Biotechnology; 2002–2009.
Csaba G, Knippel B, Karabélyos C, Inczefi-Gonda A, Hantos M, Tekes K. Impact of single neonatal serotonin treatment (hormonal imprinting) on the brain serotonin content and sexual behavior of adult rats. Life Sci. 2003 Oct 10;73(21):2703–2711.
Davidson RJ, Putnam KM, Larson CL. Dysfunction in the neural circuitry of emotion regulation – a possible prelude to violence. Science. 28 July 2000:591; doi:10.1126/science.289.5479.591.
Guyton A. Textbook of Medical Physiology. 10th ed. W. B. Saunders Company; 2000.
Hull EM, Muschamp JW, Sato S. Dopamine and serotonin: influences on male sexual behavior. Physiol Behav. 2004;83(2):291–307.
Johansson AK, Bergvall AH, Hansen S. Behavioral disinhibition following basal forebrain excitotoxin lesions: alcohol consumption, defensive aggression, impulsivity and serotonin levels. Behav Brain Res. 1999 Jul;102(1–2):17–29.
Linnoila M, Virkkunen M, Scheinin M, Nuutila A, Rimon R, Goodwin FK. Low cerebrospinal fluid 5-hydroxyindoleacetic acid concentration differentiates impulsive from nonimpulsive violent behavior. Life Sci. 1983 Dec 26;33(26):2609–2614.
Moll J, de Oliveira-Souza R, Eslinger PJ, et al. The neural correlates of moral sensitivity: a functional magnetic resonance imaging investigation of basic and moral emotions. J Neurosci. 2002 Apr 1;22(7):2730–2736.
Orten J, Neuhaus O. Human Biochemistry. 10th ed. St. Louis: C. V. Mosby Co.; 1982.
Pattichis K, Louca LL, Jarman J, Sandler M, Glover V. 5-Hydroxytryptamine release from platelets by different red wines: implications for migraine. Eur J Pharmacol. 1995 Jan 13;292(2):173–177.
Raine A. The Psychopathology of Crime: Criminal Behavior as a Clinical Disorder. Academic Press; 1993.
Rausch JL, Johnson ME, Corley KM, et al. Depressed patients have higher body temperature: 5-HT transporter long promoter region effects. Neuropsychobiology. 2003;47(3):120–127.
Shils M, Olson J, Shike M. Modern Nutrition in Health and Disease. 8th ed. Pennsylvania: Lea & Febiger; 1994.
Shufflebotham J, Hood S, Hendry J, et al. Acute tryptophan depletion alters gastrointestinal and anxiety symptoms in irritable bowel syndrome. Am J Gastroenterol. 2006 Nov;101(11):2582–2587. Epub 2006 Oct 4.
van Lelyveld N, Ter Linde J, Schipper ME, Samsom M. Regional differences in expression of TPH-1, SERT, 5-HT(3) and 5-HT(4) receptors in the human stomach and duodenum. Neurogastroenterol Motil. 2007 May;19(5):342–348.
Van Winkle E. The toxic mind: the biology of mental illness and violence. Med Hypotheses. 2000 Jan;54(1):146–156; corrected in Med Hypotheses. 2000 Oct;55(4):356–368.
Yeh SY, Dersch C, Rothman R, Cadet JL. Effects of antihistamines on 3,4-methylenedioxymethamphetamine-induced depletion of serotonin in rats. Synapse. 1999;33:207–217.
©2009 John A. Allocca, DSc, PhD |



![]()
![]()
![]()







