Shortly after the combined therapy arm of the Women's Health
Initiative (WHI) Study (which concluded that Prempro1 increases a woman's
risk of breast cancer, stroke, and cardiovascular disease) was halted,
a group of women's health experts were interviewed on Seattle's
public radio station. The experts were unanimous: "estrogens
cause cancer, and women shouldn't take them." If only it
was that simple! Since then, new research has appeared that calls many
of our initial impressions from the WHI trial into question. Some of
these questions are:
1. How much of an increase in adverse outcomes was associated with
Prempro use? The base rates of breast cancer, stroke and myocardial
infarction were low in the WHI Trial. Thus the relatively large percentage
increases in these outcomes implies only a small number of additional
cases. As one reporter put it, "Last year, 6 million post-menopausal
American women woke up to the front-page news that their hormone replacement
treatment was increasing their chances of getting breast cancer by
26%, stroke by 41% and heart attacks by 29%. What the percentage changes
actually represented, though, was a change in a rate—an additional
seven to eight women per 10,000 were diagnosed with these disorders….
Death rates were not increased at all."2
2. Which component of Prempro caused the adverse outcomes? Or was it
the combination? Some of the larger post-WHI studies are summarized
in Table 1. Although some of the trials suffer methodologically because
they were not placebo controlled, they have the strength of large numbers.
A study of 3663 women from Sweden found a large percentage increase
in breast cancer cases in women who took estrogen + progestin continuously
(as the women in the WHI Trial did). Women who took estrogen + progestin
sequentially (to mimic the natural menstrual cycle) had a smaller increase
in breast cancers, which did not reach statistical significance. Women
who took estradiol alone had no increase in breast cancer risk.3
Table 1. Some Recent Studies of Hormone Replacement Therapy
| Country |
# Women Studied |
HRT Regimen |
Effect on Breast Cancer Risk |
Statistically significant difference? |
| Sweden3 |
3663 |
Estrogen + progestin, continuous |
2-3 fold increase |
Yes |
| |
|
Estrogen + progestin, sequential |
Slight increase |
No |
| |
|
Estradiol only |
Slight decrease |
No |
| |
|
Estriol only |
Slight increase |
No |
| |
|
|
|
|
| France4 |
1739 |
Estradiol + progesterone |
No increased risk |
|
| |
|
|
|
|
| U.S.5 |
1982 |
Estrogen + progestin |
70% increase |
Yes |
| |
|
Estrogen only |
No increased risk |
|
| |
|
|
|
|
| Britain6 |
1,084,110 |
Estrogens + progestins |
100% increase |
Yes |
| |
|
Conjugated equine estrogens only |
29% increase |
Yes |
| |
|
Estradiol only |
24% increase |
Yes |
| |
|
|
|
|
| U.S.7 |
11,000 |
Estrogens only |
No increased risk |
|
A study of 1739 women from France concluded: "[t]he results
of the present study, in long-term users of a combination of transdermal
estradiol and a progestin closer to progesterone than MPA [medroxyprogesterone
acetate, used in the WHI], do not show any increase in breast cancer
risk."4
A study from the Fred Hutchinson Cancer Research Center in Seattle
found increased risk of breast cancer for women taking estrogen + progestin,
but no increased risk for women taking estrogen alone.5
The largest study to date, the aptly named Million Women Study from
Britain,6 described a gradient of breast cancer risk, with estrogen
+ progestin associated with the greatest increase in breast cancer
risk, and estrogen alone associated with a smaller, but statistically
significant, increase in breast cancer risk.
Finally, the estrogen-only arm of the WHI trial was halted in early
March 2004.7 The study concluded that, "with an average of nearly
7 years follow-up completed, estrogen alone does not appear to affect
. . . heart disease. . . . At the same time, estrogen alone appears
to increase the risk of stroke and decrease the risk of hip fracture.
It has not increased the risk of breast cancer…." (Emphasis
added).
One of the confounding factors in the studies cited is that the women
who received only estrogen had in almost all cases had their uterus
removed, and were thus sampled from a different population than women
receiving combined HRT. It is possible that there is a difference between
women with intact uteruses and women without them that confers additional
breast cancer risk. Nevertheless, at present the evidence strongly
suggests that combined HRT with a synthetic progestin increases breast
cancer risk much more than estrogen-only HRT. Evidence regarding estrogen-only
HRT is mixed: some studies show no increased breast cancer risk, but
the largest, most statistically powerful study finds a small but significant
increase in risk with estrogen alone.
3. This leads us to the next important question: Are bioidentical estrogens
safer than synthetic or equine estrogens? One of the tragedies of the
WHI trial is that the only active treatments offered were Prempro and
Premarin. Without head-to-head comparisons of bioidentical HRT formulations
and Prempro/Premarin, it is not possible to state definitively that
bioidentical HRT is safer. There is ample evidence that estrogens,
at least under certain conditions, are capable of inducing mutagenic
and genotoxic changes.8 Critical factors include the estrogen dose,
the patient's individual metabolic profile, and the relative
amounts of various estrogens present. But the magnitude of estrogen-related
carcinogenesis is relatively small: up to a 29% increase in breast
cancer risk with estrogens alone compared to roughly a 1000% increase
in lung cancer risk for smokers compared to non-smokers.
In order to reconcile and unify the clinical studies that have appeared
to date, we need to add one more piece to the puzzle: the effect of
estrogen dose. It is widely assumed that adverse effects of estrogens
are dose-related. The original recommended dose of Premarin (the conjugated
equine estrogen component of Prempro) was 1.25 mg. Subsequently; it
was lowered to 0.625 mg (the dose used in the WHI trial). Recently,
a 0.3 mg formulation was approved (although enlightened practitioners
experimented with these lower doses as far back as the 1980s9). The
same pattern is evident for estradiol. Early formulations contained
2 mg, which was subsequently lowered to 1 mg. Dr. Jay S. Cohen recommended
0.5 mg in 2001.10 Last year, a study was published in JAMA demonstrating
beneficial effects on bone health with an estradiol dose of 0.25 mg.11
What dose of estradiol replaces estrogens at normal premenopausal ranges?
The answer to this question is complicated by the fact that the interconversion
between estradiol and estrone is shifted toward estrone after menopause.
And elevated estrone concentrations are associated with an increased
risk of breast cancer.12 In Figure 1, I have plotted the urinary estrone
output for 22 post-menopausal women whose hormones were tested at Meridian
Valley Laboratory, 19 of whom were receiving estradiol orally in various
doses. Note that at estradiol doses above 0.25 mg, urinary estrone
output is uniformly above normal premenopausal levels. At estradiol
doses of 1 mg, urinary estrone output is roughly four times higher
than the upper limit for normal pre-menopausal women.
Figure
1
Urinary Estrone Output in Postmenopausal Women Taking Oral
Estradiol
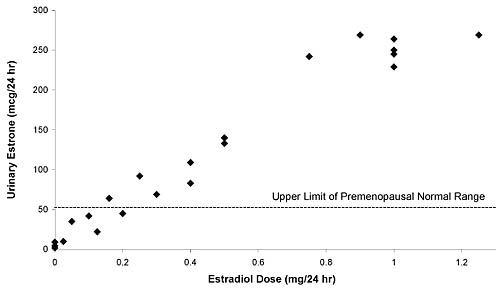
Figure 1 only shows evidence for excessive estrone in urine. What
do blood tests tell us? In many cases, only serum estradiol is
measured,
so the rate of conversion to estrone is unknown. But where this
information is available, it indicates elevated serum estrone in
post-menopausal
women receiving high-dose estrogen replacement therapy. The package
insert for one product13 that provides 1 mg of estradiol
indicates that the mean peak serum estradiol level is around 100
pg/ml, while
the mean peak estrone level is around 500 pg/ml. For comparison,
the median estrone levels in premenopausal women measured by one
popular
radioimmunoassay are: follicular phase 62 pg/ml, luteal phase 65
pg/ml, and peri-ovulatory 123 pg/ml. The package insert for this
methodology
gives expected estrone values in post-menopausal women receiving
estrogen replacement therapy (probably conjugated equine estrogens
0.625 mg)
of up to 346 pg/ml,14 much higher than the normal premenopausal
levels.
This may help us understand some of the differences in Table 1.
The French study that found no increase in breast cancer risk with
estradiol,
dealt with women receiving low-dose transdermal estradiol. Clearly,
we can expect the best results when estradiol is given at lower
doses that result in more physiological estrone serum and urine
concentrations.
It seems that those who set the original recommended doses at supra-physiological
levels may have done a great disservice to post-menopausal women.
In summary, the factors known to be associated with the most adverse
outcomes in estrogen replacement therapy are:
1) use of synthetic
progestins in combinations with estrogens,
2) use of continuous rather
than sequential estrogen administration, and
3) excessive estrogen
dose. The main arm of the WHI trial managed to combine all three
in a worst-case scenario that, not surprisingly, gave negative
results.
More research on bioidentical hormone replacement is urgently needed.
There is compelling laboratory evidence that natural progesterone
is safer than synthetic progestins.15,16 Clinical trials
are needed to confirm this. Also high on the research agenda are
oral and transdermal
low-dose estradiol, as well as estriol alone and in combination
with estradiol. Finally, we must develop a better understanding
of laboratory
monitoring of HRT—what to measure, in which body fluids, and
when to measure it. 
Correspondence:
Patrick Friel
801 SW 16th Street, Suite 126
Renton, Washington 98055 USA
425-271-8689

References
1. Conjugated equine estrogens plus the synthetic progestin medroxyprogesterone
2. Eisenstadter I. Countdown to ignorance. Barron's December
1, 2003, p. 35.
3. Olsson HL, Ingvar C, Bladstrom A. Hormone replacement therapy containing
progestins and given continuously increases breast carcinoma risk in
Sweden. Cancer. 2003 Mar 15;97(6):1387–92.
4. de Lignieres B, de Vathaire F, Fournier S, Urbinelli R, Allaert
F, Le MG, Kuttenn F. Related Articles, Links Abstract Combined hormone
replacement therapy and risk of breast cancer in a French cohort study
of 3175 women. Climacteric.
2002 Dec;5(4):332–40.
5. Li CI, Malone KE, Porter PL, Weiss NS, Tang MT, Cushing-Haugen KL,
Daling JR. Related Articles, Links Abstract Relationship between long
durations and different regimens of hormone therapy and risk of breast
cancer. JAMA. 2003 Jun 25;289(24):3254–63.
6. Beral V; Million Women Study Collaborators. Related Articles, Breast
cancer and hormone-replacement therapy in the Million Women Study. Lancet. 2003 Aug 9;362(9382):419–27.
7. http://www.nlm.nih.gov/databases/alerts/estrogen_alone.html
8. Friel PN. Laboratory Evaluation of Estrogen Metabolism. TLƒDP 245:62–66; December 2003.
9. Wright JV, personal communication.
10. Cohen JS. Over Dose: The Case Against the Drug Companies.
New York: Penguin Putnam (2001).
11. Prestwood KM, Kenny AM, Kleppinger A, Kulldorff M. Related Articles,
Links Abstract Ultralow-dose micronized 17beta-estradiol and bone density
and bone metabolism in older women: a randomized controlled trial. JAMA. 2003 Aug 27;290(8):1042–8.
12. Friel PN, ibid.
13. Activella, Physicians Desk Reference 2002.
14. Estrone RIA, DSL-8700, Diagnostic Systems Laboratories, Inc.
1999.
15. Wright JV, Morgenthaler J. Natural Hormone Replacement for
Women Over 45. Smart Publications (1997).
16. Thomas T, Rhodin J, Clark L, Garces A. Progestins initiate adverse
events of menopausal estrogen therapy. Climacteric. 2003
Dec;6(4):293–301.
|





