|
Page 1, 2, 3, 4, 5
Many patients with bloating, abdominal pain, constipation, or diarrhea are diagnosed with irritable bowel syndrome and never get adequate responses to treatment. Others are given no diagnosis at all for their suffering, which leads to even less chance of recovery. Our experience is that many of these perplexing patients have commensal microbial overgrowth. This article details the complex issue of small intestine bacterial overgrowth (SIBO)
SIBO is a condition in which abnormally large numbers of commensal bacteria (or other microorganisms) are present in the small intestine. SIBO is a common cause of IBS – in fact it is involved in over half the cases of IBS and as high as 84% in one study using breath testing as the diagnostic marker.2 It accounts for 37% of cases when endoscopic cultures of aerobic bacteria are used for diagnosis.3 Eradication of this overgrowth leads to a 75% reduction in IBS symptoms.4 Either bacterial overgrowth or the overgrowth of methanogenic archaea leads to impairment of digestion and absorption and produces excess quantities of hydrogen, hydrogen sulfide, or methane gas. Hydrogen and methane are not produced by human cells but are the metabolic products of fermentation of carbohydrates by intestinal organisms. When commensals (oral, small intestine, or large intestine flora) multiply in the small intestine to excessive numbers, IBS is likely. Hydrogen/methane breath testing is the most widely used diagnostic method for this condition. Stool analysis has no value in diagnosing SIBO.
Symptoms of SIBO include:
- bloating/abdominal gas
- flatulence, belching
- abdominal pain, discomfort, or cramps
- constipation, diarrhea, or a mixture of the two
- heartburn
- nausea
- malabsorption: steatorrhea; iron, vitamin D, vitamin K, or B12 deficiency with or without anemia; and osteoporosis5
- systemic symptoms: headache, fatigue, joint/muscle pain, and certain dermatology conditions
Other diseases associated with SIBO include hypothyroidism, lactose intolerance, gallstones, Crohn's disease, systemic sclerosis, celiac disease, chronic pancreatitis, diverticulitis, diabetes with autonomic neuropathy, fibromyalgia and chronic regional pain syndrome, hepatic encephalopathy, non-alcoholic steatohepatitis, interstitial cystitis, restless leg syndrome, acne rosacea, and erosive esophagitis.6–21 Based on clinical experience, we suspect that biliary dyskinesia and lymphocytic colitis may also be associated with SIBO.
In our practices we have found that the following indicators increase the chances that a patient's IBS is caused by SIBO:
- when a patient develops IBS following a bout of acute gastroenteritis (postinfectious IBS);
- when a patient reports dramatic transient improvement in IBS symptoms after antibiotic treatment;
- when a patient reports worsening of IBS symptoms from ingesting probiotic supplements that also contain prebiotics;
- when a patient reports that eating more fiber increases constipation and other IBS symptoms;
- when a celiac patient reports insufficient improvement in digestive symptoms even when carefully following a gluten-free diet;
- when a patient develops constipation type IBS (IBS-C) after taking opiates;
- when a patient has a chronic low ferritin level with no other apparent cause;
- when abdominal imaging reveals a large gas accumulation obscuring the pancreas
- when small bowel follow-through imaging reveals areas of "flocculation."22
Mechanisms by Which Overgrowth Is Prevented
An important protective mechanism against SIBO is proper small intestine motility via the migrating motor complex because stasis promotes bacterial growth.23 Also key in prevention are gastric, pancreatic, and gall bladder secretions, since hydrochloric acid, enzymes, and bile are bactericidal/static.24 Conditions that disrupt the glycocalyx and microvillus portions of the brush border may fuel overgrowth. The pathophysiology involved is the loss of disaccharidases in these areas and the resulting carbohydrate malabsorption which provides excess substrate for microbial growth. The role of proper ileocecal valve function in preventing cecoileal reflux of colonic bacteria into the small intestine may also be important.25,26 Surprisingly, a recent study suggests that surgical removal of the gall bladder reduces the risk as well.27 Mucosal biofilms may be preventive or may be a risk.28,29 Heavy drinking, as well as moderate use of alcohol, is significantly associated with increased SIBO risk.30 The use of proton pump inhibitors encourages overgrowth, especially of the hydrogen-producing type.31,32
Definition
Traditionally, ≥105 colony-forming units (CFU) per mL of proximal jejunal aspiration has been the definition of SIBO in culturing studies. ≥103 CFU is now the suggested definition from more recent studies revealing that ≤103 CFU is the normal level in healthy controls.33,34 The bacteria which are most commonly overgrown are both commensal anaerobes – Bacteroides 39%, Lactobacillus 25%, Clostridium 20% – and commensal aerobes – Streptococcus 60%, Escherichia coli 36%, Staphylococcus 13%, Klebsiella 11%.35 A more recent study found the aerobes to be Escherichia coli 37%, Enterococcus spp 32%, Klebsiella pneumonia 24%, and Proteus mirabilis 6.5%.36 Colonic hydrogen production is believed to be anti-inflammatory and antineoplastic, whereas excessive small intestine hydrogen causes the symptoms and signs of diarrhea-type irritable bowel syndrome (IBS-D).37 In addition to bacteria, the source of methane generation in SIBO is the archaeon Methanobrevibacter smithii. This organism has been linked to obesity in humans.38 In addition, sulfate-reducing bacteria, such as Desulfovibrio species, are anaerobes that reduce sulfate to hydrogen sulfide (H2S). In addition to its role in SIBO, H2S is being studied as a possible etiologic factor in ulcerative colitis and colonic carcinogenesis.39 In normal low levels H2S has GI protective activity.40
Pathophysiology of SIBO: Autoimmunity
Postinfectious IBS (PI-IBS) has been shown to have an autoimmune etiology in both murine and human studies (see Figure 1). Infectious gastroenteritis is the most significant environmental risk factor for IBS.41 Organisms that trigger PI-IBS include Campylobacter, Salmonella, Shigella, E. coli, viruses, and Giardia.42–45
Figure 1
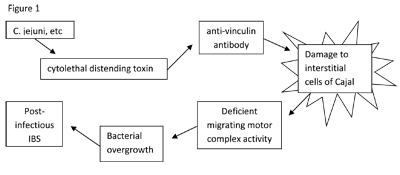
Cytolethal distending toxin (CDT) is produced by enteric pathogens that cause PI-IBS. Campylobacter jejuni is the prototypical bacterium that produces CDT.46 Other bacteria that produce CDT include Haemophilus ducreyi (chancroid), Aggregatibacter actinomycetemcomitans (periodontitis), Escherichia coli (traveler's diarrhea), Shigella dysenteriae (dysentery), Salmonella enterica (typhoid fever), and Campylobacter upsaliensis (enterocolitis).
The interstitial cells of Cajal (ICC) are fibroblast-like cells that act as pacemakers for the migrating motor complex (MMC). A key underlying cause of SIBO is thought to be deficiency of the MMC, which moves debris and bacteria down into the large intestine during fasting at night and between meals.47 The number of ICC is reduced in post-Campylobacter jejuni gastroenteritis infected rats that eventually develop SIBO.48 Three months after C. jejuni gastroenteritis, 27% of rats had SIBO. These rats had a lower number of ICC than controls in the jejunum and ileum (0.12 ICC/villus was the threshold for developing SIBO).
CDT toxin may destroy the interstitial cell of Cajal by stimulating the production of autoantibodies against a cytoskeletal protein known as vinculin. The antigen-antibody complexes between antivinculin antibodies and cytolytic distending toxin lead to autoimmune destruction of ICC.49,50
How SIBO Causes the Symptoms of IBS
There are two main pathophysiological issues involved in SIBO. First, bacteria can ferment carbohydrates and consume other nutrients ingested by the host simply by their inappropriate location in the small intestine. This allows them premature exposure to host nutrition before there is time for absorption. Bacterial fermentation produces hydrogen and/or hydrogen sulfide gas. In addition M. smithii produces methane.51 M. smithii may be present in the intestinal tracts of up to 95.7% of humans.52 Microbial gas leads to the IBS symptoms of bloating, pain, altered bowel movements, eructation, and flatulence.
Figure 2
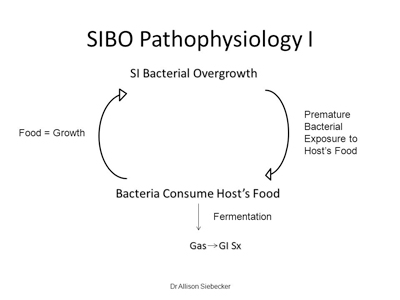
The quantity of gas may be extensive, causing severe bloating and distention.53 It is estimated that with normal levels of enteric flora, the quantity of lactose in an ounce of milk fuels the production of 50 cc of gas. With microbial overgrowth, gas levels produced from 1 ounce of milk may approach 5000 cc.54 Excess gas can then exit the body as flatulence or eructation. A portion is also absorbed into the blood and eventually filters through the pulmonary alveolus to exit on exhalation. The intestines are sensitive to pressure, and therefore the pressure of distention can lead to abdominal pain. In addition, visceral hypersensitivity, a feature of IBS, may create a lower threshold for pain/discomfort and a hyperresponsiveness of muscular contraction in response to the gas, leading to cramps.55,56 The gases also affect bowel motility. Hydrogen has a greater association with diarrhea, and methane has an almost exclusive association with constipation.57,58 Methane has been shown to slow gastrointestinal motility by 59% in animal studies, and the volume of methane overproduction correlates with the severity of constipation.59,60 Therefore when both hydrogen and methane are present, diarrhea, constipation, or a mixture of both can be present based on the relative amounts of these gases.61 It appears that the pressure created by either gas or the decreased gastric motility may lead to gastric distention resulting in gastroesophageal reflux (GERD).62 The bacterial consumption and uptake of host nutrients, such as B12 and iron, can lead to macrocytic and/or microcytic anemia or chronic low ferritin levels in addition to general malabsorption and malnutrition in more severe cases.63,64 The increased motility of diarrhea may also induce malabsorption. Finally, continuous fermentation of host nutrition by repeated exposure to daily meals perpetuates bacterial overgrowth and IBS symptoms, creating a vicious cycle (Figure 2).
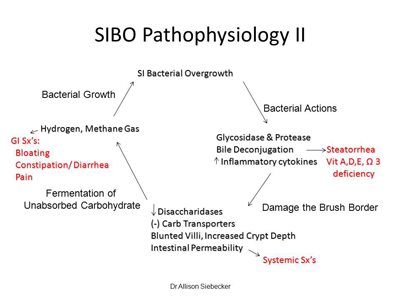
The second mechanism is microbial damage to the digestive and absorptive function of the small intestine. Unlike the colon, the small intestine is not designed for heavy bacterial colonization. Commensal organisms may synthesize glycosidase, leading to damage of glycocalyx or disaccharidases. The gastrointestinal and systemic symptoms induced by these changes are listed in Figure 3. Key factors include bacterial deconjugation of bile, which induces fat malabsorption, steatorrhea, and fat–soluble vitamin deficiencies; bacterial digestion of disaccharidase enzymes, which furthers carbohydrate malabsorption, fermentation, and gas; and increased intestinal permeability (leaky gut), which often leads to systemic symptoms.65–68
Figure 3
Page 1, 2, 3, 4, 5
|
![]()
![]()
![]()







