|
Page 1, 2, 3
There has been some confusion expressed on the Internet regarding the oxalate content in green smoothies along with a "dire warning" (in at least one article) to avoid drinking liquid sunshine.23
This sort of confusing rhetoric is intended to bewilder and baffle people rather than to enlighten them, and is always to be expected whenever a true healing modality directly from the loving hands of Nature (and nonpatentable) gains in popularity. And, although the compassionate intention of these commentaries to help mitigate the occurrence of calcium-oxalate kidney stones is appreciated, glaringly absent from these admonitions are warnings for people to avoid or at least reduce their intake of animal protein and refined carbohydrates which, for the population at large, should be of much greater concern than green smoothies.
Four Types of Kidney Stones
Four major types of kidney stones can form:
- Calcium stones are the most common and occur in two major forms: calcium phosphate and calcium oxalate.
- Uric acid stones result when the urine is consistently acidic. Eating purine rich food (animal protein) can result in calcium urate stones (or uric acid stones).
- Struvite stones are associated with kidney infections.
- Cystine stones are a consequence of a specific genetic disorder.
Oxalates in Human Food
There are many foods commonly found in the human diet that contain oxalates in significant concentrations including the following:
- Fruits
° Mafuang (starfruit or carambola), berries, currants, kiwifruit,
Concord (purple) grapes, figs, plums, and tangerines.
- Vegetables
° Parsley, beet greens and roots, spinach, Swiss chard, collard greens,
okra, leeks, celery, and rutabagas.
- Seeds and nuts
° Pumpkin, squash, sunflower, poppy, quinoa (a seed), almonds,
cashews, macadamia, filberts, etc.
- Legumes
° Soybeans (tofu) and peanuts
- Grains
° Wheat bran, wheat germ, and buckwheat
- Other
° Cocoa, coffee, chocolate, and green and black tea.
Note: leaves contain higher concentrations of oxalates than do stems, stalks, and roots.
What are Oxalates?
Oxalates are the salts of oxalic acid, which occur naturally in many plants as a product of metabolism.
Salts (ions/charged) result from neutralizing acids and consist of positively charged cations in association with negatively charged anions in a solution, such as water. There needs to be a certain amount of water (solute) to keep the salt in its ionic form. Below a certain threshold amount of water, the cations and anions come together (precipitate) to form crystals (e.g., table salt).
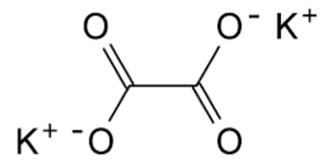
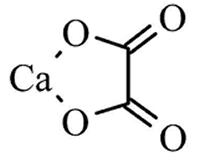
Oxalates are divalent anions, which means that they can "grab" two monovalent cations, such as two atoms of potassium (see Figure 1), or one divalent cation, such as calcium, iron, magnesium, and so on (see Figure 2).
Figure 1: K2 Oxalate
|
Figure 2: Calcium Oxalate
|
How Do Oxalates Produce Kidney Stones?
When ingested oxalates that are not bound by cations in the gut (e.g., calcium) can be absorbed directly into the blood and travel to the kidneys, where if the conditions are suitable and there is sufficient calcium present, calcium-oxalate crystals form. Whether the oxalate and calcium remain in ionic form or crystalize (precipitate) depends upon several factors, including pH (acidity), water content (hydration status), and the presence of other nutrients.
The point at which precipitation (crystallization) occurs is known as the saturation point and depends on a multitude of factors other than the oxalate content of food.*
In fact, the oxalate content of food actually has little relevance as to whether oxalate stones will form in the kidneys.
There are multiple studies on rats and humans indicating that high dietary intake of calcium actually reduces the incidence of oxalate kidney stones and, furthermore, that magnesium and potassium intake are inversely related to oxalate stone formation.
Hence, the more calcium, magnesium, and potassium that are in the diet, the more the ingested oxalates will be bound (in the intestines) and thereby excreted through the bowels rather than reabsorbed into the blood and subsequently excreted through the kidneys where they could precipitate to form crystals (stones). Additionally, the presence of adequate magnesium in the blood greatly reduces the likelihood of calcium-oxalate stone formation.
*The solubility (dissolvability) of oxalates varies greatly depending upon the cations present. The most soluble (dissolvable) are magnesium (Mg) and calcium (Ca) and the least soluble are the "heavy metals," such as lead (Pb) and mercury (Hg). Therefore, when oxalates are bound to magnesium or calcium, they are more likely to remain dissolved and not crystalize, whereas when bound to heavy metals, they crystalize quite readily.
Dietary vs. Supplemental Calcium
A very important distinction needs to be emphasized between true dietary minerals (e.g., calcium) and supplemental calcium in pill or powder form. It has been shown that although high intake of dietary calcium decreases the risk of kidney stones, the intake of supplemental calcium can actually increase the risk of stone formation depending upon timing and amount.
The metabolic pathway (see Figure 3) represents the endogenous production (made by the body) of oxalates and clearly shows that kidney stones are related to dietary protein and refined carbohydrates, not to the oxalate content of food.
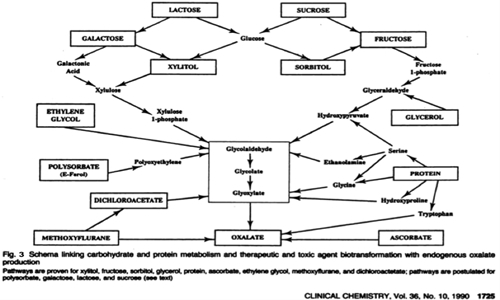
Furthermore, some foods that have quite high oxalate contents such as black and green tea actually appear to be preventative with regard to oxalate stone formation.
A very interesting study published in 1998 in the Annals of Internal Medicine evaluated just over 81,000 women aged 40 to 86 years who had no history of kidney stones. The results were quite confusing to these researchers, since for each cup of green tea consumed on a daily basis, the women's risk of kidney stones was actually reduced by 8%. In another study involving men, it was found that for each cup of tea consumed, their risk of kidney stones was reduced by 14%.
How Important are Dietary Oxalates?
It is well known that 80% to 90% of oxalates excreted in the urine are endogenously produced (made in the body), thereby decreasing the potential role of dietary oxalates to between 10% and 20%.4,5,7,8
Since dietary oxalate intake accounts for such a small amount of the oxalates actually found in the urine of people who form calcium oxalate stones, it is now fairly well accepted that dietary restriction of oxalate-containing foods is not a viable therapeutic intervention to prevent stone formation, except in a few rare circumstances: hypercalciuria type II and hyperoxaluria (primary and enteric).4,5,7,8,16,21
However, even in conditions involving fat malabsorption or inflammatory bowel disease (enteric hyperoxaluria), if probiotics are taken daily for 2 months, the saturation of the urine is reduced to such an extent that approximately 25% less calcium related (oxalate) stones are formed.
When any of these conditions do exist, however, the recommendation is that only about one-fourth cup of spinach or its equivalent should be consumed per day. That still leaves plenty for many smoothie recipes.
What Causes Kidney Stones?
Recent research indicates that the intake of refined carbohydrates, protein, calcium, and water are much more relevant to calcium oxalate stone formation than dietary oxalates.
The British Association of Urological Surgeons published the following, which is fully agreed on by the international medical community in all medical specialties: "Dietary advice to increase the consumption of fibre and reduce the consumption of sugar, refined carbohydrates and animal protein produced a significant reduction in the urinary excretion of calcium, oxalate and uric acid."
Animal Protein
Animal based diets generate large amounts of acid in the various fluid departments of the body, hence the kidneys respond by excreting all excess acids to maintain an alkaline pH in the blood and ECM (fluid bathing the cells).
Calcium, in the form of calcium bicarbonate (Ca[HCO3]2), calcium carbonate (CaCO3), and calcium phosphate (CaPO4), is released from the bones (in order to neutralize the excess acid), where it enters the blood, the extracellular fluid (ECM), and through the kidneys, thereby increasing the amount of urinary calcium available to precipitate with oxalates to form stones. This homeostatic response by the body to maintain functional integrity, however, results in a net calcium loss from the bones (osteoporosis).11,17-19,21
In other words, calcium is leached from the bones to neutralize the acids formed by consuming animal protein, resulting in supersaturation of calcium in the urine available for stone formation and less calcium in the bones (osteoporosis).11,17-19,21
Additionally, animal protein is the major dietary source of purines, which are broken down into uric acid that leads to uric acid kidney stones and excruciatingly painful crystals in joints (gout).
Of great import, it should be noted that dietary vegetable protein consumed in high amounts does not contribute to these same pathological changes in uric acid metabolism and calcium metabolism, hence does not lead to gout, osteoporosis, kidney stones, or kidney failure, as do large amounts of animal protein.
"A study conducted in the UK showed that a diet low in animal protein reduced the prevalence (occurrence) of urinary stone formation by 40–60%"17
"A high animal protein intake causes a significant increase in the urinary excretion of calcium, oxalate, and uric acid, 3 of the 6 main urinary risk factors for calcium stone formation."16
"High amounts of dietary protein can lead to increases in both calcium and oxalate levels in the urine. The elevated protein results in lower urine pH – an acidic environment that makes it easier for calcium oxalate kidney stones to form. It also decreases citrate levels in the urine that help prevent kidney stones from forming. …"9,15,16
Page 1, 2, 3
|
![]()
![]()
![]()







