|
Part 1, Part 2 and Part 3 are also online.
Mirko Beljanski's Products for Cancer Patients
Four Supplements to Help Support Cancer Patients
Four supplements that I have used extensively in my practice for cancer patients since 1999 were developed by the late Mirko Beljanski, PhD, a molecular biologist who was born in Yugoslavia, but spent most of his adult life doing biological research in France. He did extensive research on RNA and DNA and developed a theory about cancer that is quite profound. Information and books about his work are available at two websites: www.beljanski.com and www.natural-source.com. I had never heard of Beljanski or his research prior to 1999; but when I was advised by a knowledgeable friend to attend a weekend conference that year concerning natural products for cancer patients, sponsored by a company named Natural Source, I decided to go. At this conference were residents of a few European countries (mostly France and Belgium) who were scientists, physicians, and cancer patients who either had benefited from these supplements developed by Beljanski or who had researched them or had administered them to patients with apparent benefits.
Dr. Mirko Beljanski's Background and Research
I learned that Beljanski had died in 1998 after being persecuted by the French government for suggesting that these natural, nontoxic supplements might be helpful to cancer patients as adjuncts to their conventional treatment. His daughter Sylvie, an attorney, started this company Natural Source in New York City to promote her father's research work and his products. I was impressed with most of the presentations at this bilingual conference (French-English), reviewed many of Beljanski's research papers, and began to use the supplements with some of my cancer patients. Many patients appeared to benefit from them. During his lifetime, Beljanski did research for 30 years at the Pasteur Institute. He published 133 papers. Most of the earlier ones were in French and the later ones were mostly in English. Much of his work with RNA and DNA is summarized in his monograph, originally published in 1983.93
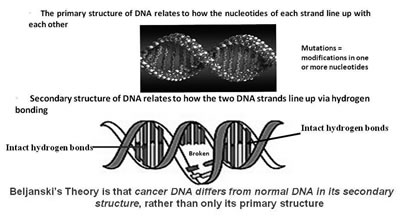
Beljanski's Cancer Theory involving the Secondary Structure of DNA
Beljanski's basic research on RNA and DNA led to the development of a theory about a mechanism for the development of cancer. During the 1960s and 1970s, the predominant theory for the development of cancer was that mutations in the primary structure of DNA in certain classes of genes; namely, pro-oncogenes, suppressor genes, and DNA repair genes, resulted in malignant changes in cells. Generally, the more mutations in these genes, the more aggressive would be the cancer cell. This change in the primary structure of DNA involved mutations, consisting of a change in one of the possible four bases, that were present in a pattern along the two DNA strands of the double-helix structure of DNA.
Beljanski's new idea was that rather than just changes in the primary structure of DNA, changes in the secondary structure of DNA might contribute to the development of cancer. The secondary structure of DNA relates to the hydrogen bonding that takes place between the two strands of DNA. He suggested that carcinogenic substances could interfere with some of the hydrogen bonds between the two strands of the double helix, causing the formation of large loops between the strands. When this occurred, the DNA became destabilized, increasing the risk of a cell becoming cancerous.94,95 This theory is illustrated nicely in Figure 7.
Figure 7
The Oncotest for Determining Carcinogenic Substances
From this theory, Beljanski developed a test, which he called the Oncotest, to determine the carcinogenicity of a substance. He found that when he added a known carcinogenic substance to a solution of cancerous DNA, the cancerous DNA would proliferate. He could measure the increased growth of the cancerous DNA by measuring the absorbance of light. A carcinogen destabilized DNA and caused it to grow. In this way, he could measure an unknown substance's carcinogenic potential.96
Anti-Carcinogenic Substances or Bolt Molecules (Extracts of Pao Pereira and Rauwolfia Vomitoria)
Having developed a test to evaluate whether or not a substance could destabilize DNA, which contributed to the development of cancer, Beljanski wondered if he could find substances that could restabilize DNA, which he termed bolt molecules. He screened many different compounds and found some herbal alkaloid substances that could do this.97
Figure 8

He extensively tested extracts from two herbs (pao pereira and Rauwolfia vomitoria, shown in Figure 8) and found that each of them had selective damaging effects against a wide variety of cancer cells in vitro, including liver, thyroid, brain, and breast. Applying these compounds to cancers in mice, he showed that agents were selective in destroying cancer cells and not harming normal cells and also that they were synergistic with at least some chemotherapy drugs.98 He found that the Rauwolfia vomitoria extract (also called BG-8) was slightly better in hormone related cancers, while pao pereira (also called PB-100) was capable of crossing the blood–brain barrier and therefore might be helpful in brain cancer.86
Figure 9
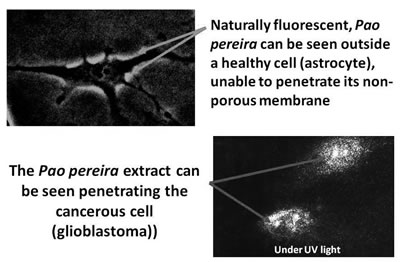
He was able to demonstrate clearly that pao pereira could penetrate and damage glioblastoma cancer cells while not penetrating normal brain glial cells (as shown in Figure 9) and had several other papers published on the effects of pao pereira on brain cancer cells.99-103 In another publication, he showed that both pao pereira (PB-100) and Rauwolfia vomitoria (BG-8) were active against human melanoma cells but not nonmalignant fibroblasts.104 Recently, Natural Source has funded research at Columbia University's Holistic Urology Department, headed by Aaron Katz, MD. One recently published paper showed that pao pereira had anticancer effects against prostate cancer cells in vitro.105 Both the pao pereira and Rauwolfia vomitoria supplements are available commercially from Natural Source and can be obtained as nutritional supplements to support cancer patients. The dosage we generally use with each of these supplements is about 2 capsules of each three times daily. Natural Source also has combinations of these extracts in one capsule.
RNA Primers to Enhance Production of Normal White Blood Cells and Platelets
In addition to his work with anticancer herbal extracts, Beljanski had two more lines of research that led to potential breakthroughs in the management of cancer patients. The first one involved enhancing normal white blood cell and platelet formation; the second will be discussed in the next section. It involves inhibiting damage from radiation. Beljanski discovered that normal cells undergoing replication require RNA entities that he called primers, which improve the efficiency of the replication process. This is shown in Figure 10.
Figure 10
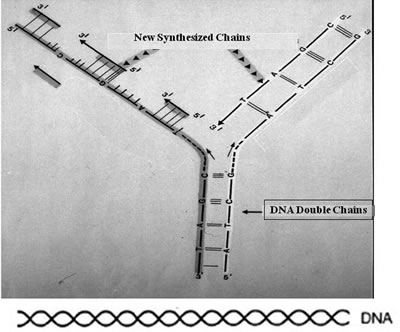
The RNA primers interact with the newly formed DNA strands to bring about enhanced replication of the DNA. He found a way to produce large quantities of these RNA primers, using a strain of nonpathogenic Escherichia coli. These RNA primers had profound effects on some of the cells in the bone marrow. In a published paper in 1979, Beljanski was able to show that, with RNA primers, he was able to drastically increase the production of all normal white blood cells and megakaryocytes that led to the production of platelets but not red blood cells in rabbits.106 Incidentally, this discovery was an extremely important breakthrough in biological understanding. Up until this time, the dogma was that DNA can affect the production of RNA, but not vice-versa. In fact, Beljanski ran into trouble at the Pasteur Institute with the director, who was one of the discoverers of how DNA affects the production of RNA. The director was angry that Beljanski insisted on publishing his own studies that questioned the dogma of the time: that DNA could affect RNA, but that the reverse was not true.
Figure 11
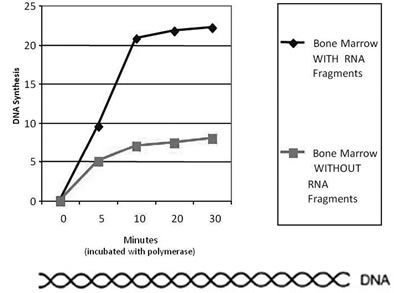
Figure 11 shows how normal DNA synthesis is increased with these E. coli RNA primers. A year later, Beljanski was able to show that these short-chain RNA fragments could serve as promoters of leucocytes and platelets in animals depleted by anticancer drugs.107 In 1981, Beljanski and Plawecki published a paper showing how the RNA primers were able to save the lives of rabbits that developed severe leucopenia following the administration of the chemotherapeutic agent cyclophosphamide. With the administration of the RNA primers, the rabbits were able to increase their leucocyte production. In this study, all of the untreated rabbits died within 10 days, while all of the treated rabbits lived for 50 days, having received the RNA primers after each dose of cyclophosphamide (See Figure 12).108,109 The rabbits that received the RNA fragments were able to create a new generation of white blood cells every time they were given a new dose. There was no exhaustion of the bone marrow. There was no release of existing immature cells. Instead, there was the creation of brand new stem cells that provided healthy white blood cells. Over the whole duration of the treatment, the rabbits were able to create a new generation of white blood cells every other day. This makes this product very different from the current approved drugs in the US, as these drugs stimulate only the production of neutrophils while the RNA primers stimulate the production of all kinds of white blood cells plus platelets.
Figure 12
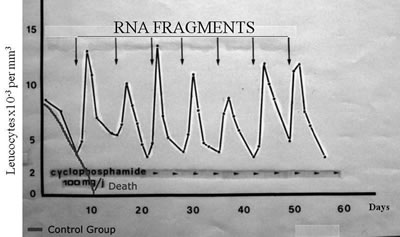
In 1991, Beljanski and his colleagues reported on the successful use of RNA fragments in two patients with non-Hodgkin's lymphoma.110 Recently, I heard a verbal report about the RNA fragments given to cancer patients presented by a PhD from the Cancer Treatment Centers of America. He said that the oncologist, with whom he had worked frequently, had to stop doing chemotherapy treatments with his patients because the patients developed dangerously low platelet counts (as many as 40% had to stop treatment). They then used the RNA fragments and none of the patients had to stop treatment. This report has not yet been published, but the preliminary results sound quite promising. The RNA fragments are available as a powder in a cone. This powder is dissolved sublingually about 3 times a week when radiation and/or chemotherapy are given. However, larger doses were used during the Cancer Treatment Centers of America's preliminary trial. Patients received the treatment as often as twice daily when the platelets dropped and apparently this worked.
One would think that all patients receiving conventional chemotherapy and radiation might benefit from treatment with these RNA primers. However, since they are not patentable drugs (as they are natural products) and the amount of money required to go through the procedures of the Food and Drug Administration (FDA) for them to become approved as a drug is astronomical for a small supplement company, it is difficult to see how they can be brought to the attention of the health-care community. At the same time, the company must be very careful not to make any claims because the FDA and/or Federal Trade Commission (FTC) could close it down for making unapproved statements for unapproved drugs. I don't know what the solution to this problem is other than to change the system or to help practitioners become aware of the benefits of these nutritional supplements and to use these safe products with their patients while they undergo conventional treatment. Practitioners will find out soon enough if the RNA primers work, and I think they will work if they are used properly.
An Herbal Extract to Help Regulate Enzymes and Reduce Abnormal Fibrosis
The final Beljanski nutritional product, potentially helpful to cancer patients, evolved from a different phase of Beljanski's research related to protection against radiation fibrosis and correction of enzyme dysregulation that occurs in many chronic conditions, including cancer. It is a specially prepared form of Ginkgo biloba that shows evidence of protection against damage from radiation and correction of enzyme dysregulation.
In the early 1990s, the French army hired Beljanski to find a way of reducing radiation damage using an American antiradiation product known as W2721. In 1991, Beljanski published a paper showing how this product, along with his RNA primers, was able to protect against radiation.111 Although W2721 was an effective protector, it had two serious drawbacks; namely, it had to be administered intravenously and it had to be kept at a very low temperature. These constraints were not compatible with the military's needs and were the reason that the product was abandoned. As allowed by the contract, Beljanski sought other alternative substances. Due to both its biochemical makeup and legendary resistance to environmental stresses such as the atomic bombings of Hiroshima and Nagasaki, Beljanski began to focus on the Ginkgo biloba tree.112
Beljanski developed a method of preparation of Ginkgo biloba quite different from the usual ginkgo preparations available commercially, and his preparation had different qualities and different actions. It was harvested at a different time of the year, which allowed him to use golden rather than the green leaves; and it was extracted with water rather than alcohol. The original name was Bioparyl but it was later renamed by Natural Source as Ginkgo V. This preparation turned out to be an enzyme regulator. As mentioned above, Beljanski showed that normal RNA primers were necessary for the replication of normal white blood cells and the formation of megakaryocytes that lead to the formation of platelets. Cancer cells require abnormal RNA primers and abnormal ribonuclease enzymes that break down RNA into these smaller chains. So, ribonucleases are very active in cancer cells. This ginkgo extract reduces ribonucleases and normalizes them in cancer and in other abnormal conditions such as autoimmune diseases. It also helps to normalize and regulate other abnormal enzyme activity. Another aspect of this enzyme regulatory activity is that this ginkgo extract was found to reduce abnormal fibrosis or scarring from radiation and from wound healing.113 The usual dose of this supplement is 2 capsules 3 times daily. Therefore, patients undergoing radiation or having undergone surgery should consider using this nutritional supplement to promote healing and prevent abnormal fibrosis.
To summarize, the research work of Mirko Beljanski led to the development of four nutritional supplements that are potentially beneficial to cancer patients either alone or in combination with conventional treatments. Two of them (extracts of pao pereira and Rauwolfia vomitoria) have selective anticancer activity, meaning that they selectively damage cancer cells but not normal cells. One of them (RNA fragments from nonpathogenic E. coli) is useful for stimulating all types of normal white blood cells and platelets and is particularly beneficial for patients undergoing radiation and/or chemotherapy. Finally, the last special extract from Ginkgo biloba has been used as a nutritional supplement to help prevent abnormal scar tissue formation from radiation or surgery.
Beljanski's Cooperative Work with Physicians and His Political and Legal Problems
As long as Beljanski's research work was confined to his laboratory, he had no legal problems with authorities. However, as colleagues and patients became aware of his research, there began to be a demand to use what he had developed to help people. His work was pretty much ignored or rejected by conventional oncologists, but some family practitioners in France and Belgium wished to incorporate the results of his work into helping their patients, and an informal network began to develop. This demand grew rapidly as many patients with cancer and other conditions greatly benefited from his products, all of which could be taken orally or sublingually (in the case of the RNA fragments). Thousands of patients became aware of his nutritional products and began to take them, often along with conventional treatments. Beljanski always wanted to work cooperatively with the medical profession and wished his products to be used with conventional treatments. Many patients, under the supervision of their health professionals, administered the plant extracts to work synergistically with surgery, radiation, and chemotherapy. Others sought out Beljanski on their own and began to take the plant extracts after being told by conventional physicians that nothing more could be done. Many of them thwarted their prognoses and wound up doing extremely well. Beljanski's reputation grew and improved.
Unfortunately, however, for a variety of reasons, including a challenge to the prevailing medical paradigm (see discussion below), a threat to the economic status quo, professional envy and jealousy, and many other factors (which involved some very powerful people), Beljanski began to develop some enemies, as always occurs when there is a threat to the current medical paradigm and threats to the economic status quo. A growing danger for Beljanski was developing.
 Perhaps the most famous patient who benefitted from the Beljanski products was the late former French President Francois Mitterrand (Figure 13). During his term in office, Mitterrand developed prostate cancer, which he kept a secret until the middle of his second term. By this time, the cancer was so advanced that his prognosis was considered grim and he was predicted to live only a few months by his conventional physicians. The country was preparing for early elections when Mitterrand decided to use the Beljanski products. Against all odds, his health began to improve and Mitterrand was able to remain in power until the end of his term. But, in the process, some powerful people became infuriated because some natural alternative treatment had thwarted Mitterrand's prognosis and thus the opportunity for some of them to seize power. So, when Mitterrand ultimately died about 18 months after he left office, the French government brutally shut down Beljanski's laboratory and mercilessly persecuted him. He was accused of practicing medicine without a license and other similar charges. Unable to do what he loved and being blocked from leaving the country, Beljanski became ill and did not have access to his own products. He died in 1998. Around that time, with a pledge to her father that she would continue his work, his daughter set up Natural Source in the US with the help of her mother, Monique, who had worked with Mirko for close to 50 years. Perhaps the most famous patient who benefitted from the Beljanski products was the late former French President Francois Mitterrand (Figure 13). During his term in office, Mitterrand developed prostate cancer, which he kept a secret until the middle of his second term. By this time, the cancer was so advanced that his prognosis was considered grim and he was predicted to live only a few months by his conventional physicians. The country was preparing for early elections when Mitterrand decided to use the Beljanski products. Against all odds, his health began to improve and Mitterrand was able to remain in power until the end of his term. But, in the process, some powerful people became infuriated because some natural alternative treatment had thwarted Mitterrand's prognosis and thus the opportunity for some of them to seize power. So, when Mitterrand ultimately died about 18 months after he left office, the French government brutally shut down Beljanski's laboratory and mercilessly persecuted him. He was accused of practicing medicine without a license and other similar charges. Unable to do what he loved and being blocked from leaving the country, Beljanski became ill and did not have access to his own products. He died in 1998. Around that time, with a pledge to her father that she would continue his work, his daughter set up Natural Source in the US with the help of her mother, Monique, who had worked with Mirko for close to 50 years.
Today, the Beljanski products are still relatively unknown to most physicians, including oncologists throughout the world. They are being recommended in Europe by a handful of clinicians and are being used primarily in France and Belgium by a minority of cancer patients. In the US, a few integrative physicians, including myself, have used them primarily with cancer patients. In my opinion, they have value and should be used as part of a total integrative cancer treatment program.
Stanislaw Burzynski MD, PhD and Antineoplastons: The Science and Legal Problems
A native of Poland with an MD and a PhD in biochemistry, Burzynski migrated to the US in the early 1970s. His research led him to the conclusion that cancer patients appeared to have a deficiency of a number of small molecules in both blood and urine that were present in higher amounts in normal patients. He observed this phenomenon in animals as well. Furthermore, these natural nontoxic substances appeared to have strong anticancer activities. He named these substances antineoplastons, and postulated that animals and humans have two defense systems in the body. The immune system primarily addressed infectious processes, while the antineoplaston system had evolved primarily to deal with cancer. He further showed that these antineoplastons work by switching certain genes on or off. A person with cancer has procancer genes (oncogenes) turned on and anticancer genes (suppressor genes and DNA repair genes) turned off. The job of the antineoplastons is to do the opposite, namely turn procancer genes off and turn anticancer genes back on. Doing basic research with cancer cells in vitro and animal experiments, Burzynski has been quite successful in accumulating evidence for his theories. Additionally, he has run clinical trials and has helped many advanced cancer patients against massive odds. His survival statistics for brain cancer in adults and children are the best in the world. He has published numerous scientific studies internationally in peer-reviewed journals. Burzynski authored an excellent review of his work along with a listing of many of his publications in the journal Integrative Cancer Therapies.114
Nevertheless, like Beljanski, Burzynski has had many problems with regulating agencies. Whereas Beljanski's legal problems occurred in France, Burzynski's problems occurred in the US. Burzynski has had problems with the FDA, the state of Texas, the Texas Medical Board, and other agencies. Unlike Beljanski in France, Burzynski has been amazingly successful in battling these regulatory agencies. A book that summarizes Burzynski's scientific and clinical work, as well as his legal trials and tribulations, is Thomas Elias's The Burzynski Breakthrough: The Most Promising Cancer Treatment… and the Government's Campaign to Squelch It.115 A documentary full-length motion picture about Burzynski's struggle with the various regulatory agencies was released in early 2010. With incredible precision and documentation, this film, Burzynski, directed by young Eric Merola, shows what Burzynski has experienced and how the government and other regulatory agencies seemingly stopped at nothing to destroy his career and his work.116 In April 2010, it won the Humanitarian Vision Award at the Newport Beach Film Festival 2010. Go to www.burzynskimovie.com and see this movie as soon as you can. It is a must. Burzynski's success has come as a result of many factors. These have included his unbelievable tenacity as a person, clinician, and scientist; his patients who have attended court hearings and congressional hearings; pressure from the US Congress (as a result of hearings) on the FDA; and many influential friends such as Julian Whitaker, MD, who has a widely read influential health newsletter and who mustered support and money for Burzynski's cause. So, with great reluctance, the FDA has allowed him to try to obtain drug approval for his products. This is the first time in history that a private clinician and scientist has been allowed to do this. The FDA's policies are such that only large pharmaceutical companies with large financial support can go through this process. So, at the time of this writing, antineoplastons can be used for only very few conditions.
On the other hand, Beljanski's work is being evaluated and used at the University of Kurume Medical School in Japan. Several papers from this university have been published over the past few years about antineoplastons.117-120
Other Nutritional Supplements
This article has barely scratched the surface on the use of nutritional supplements to support cancer patients. I have chosen to emphasize certain supplements that have been very controversial (most of the ones I discussed), not widely known among clinicians, or that I thought could be implemented immediately to help patients. This is not to say that there aren't many, many other nutritional supplements for which there is a great deal of evidence for benefits. These are seen in the books of Boik and Quillin discussed in previous sections of this article, the Jaakkola study on small cell lung cancer, and the Hoffer studies also previously discussed. Among the areas that really deserve much more attention are the systemic use of proteolytic enzymes (as utilized by the late Donald William Kelly and Nicholas Gonzalez and written about by Ralph Moss); the use of high doses of vitamin D3 to produce optimal serum levels; the use of vitamin K to help with utilization of vitamin D; vitamin A; various phytonutrients such as sulforaphane from broccoli extracts; resveratrol; curcumin; various pre- and probiotics; and many, many others. The trick is to try to put all of this together in a comprehensive manageable program for the cancer patient. To cover these in depth would indeed require a book and some of this material is available in the books and articles that have been cited.
Old vs. New Paradigms in Health Care and the Politics and Economics of Health Care Worldwide
Finally, it is necessary to touch on the difficulties of trying to practice integrative oncology and/or integrative medicine in general in the current international environment. Whitaker, an integrative physician with his own newsletter, a great following, and a major supporter of Burzynski both financially and in many other ways, appears in the movie Burzynski. He tries to explain the resistance to new ideas in health care, and his explanation was an eye-opener to me, since my own thoughts were that the problem is entirely related to economics. However, this does not explain it all. Whitaker points out that it is very hard to change a paradigm, and the paradigm for health care is very rigid.
He uses as an example the case of Dr. Ignaz Philippe Semmelweis, a Hungarian physician described as the "savior of mothers," who discovered by 1847 that the incidence of puerperal fever could be drastically cut by the use of hand disinfection (by means of hand washing with chlorinated lime solution) in obstetrical clinics. Puerperal fever, which was discovered later to be due to a bacterial infection caused by physicians moving from pathology laboratories where bodies were dissected to obstetric wards, killed many women and their offspring (mortality estimated at between 10% and 35%). Rather than greeting him with awards, colleagues of Semmelweis belittled him and failed to follow his advice, resulting in continued deaths. It was not until Pasteur explained Semmelweis's recommendations in terms of germ theory that they were implemented many years after his death. Semmelweis died a pauper in a hospital from a psychiatric or neurological disease at the age of 47 in 1865, 18 years after first offering his theory and recommendations. Whitaker points out that in Semmelweis's situation, there was no money at stake but only the difficulty changing the medical paradigm.
Today, the conventional medical paradigm involves a doctor, collecting signs and symptoms from a patient, possibly ordering some tests, coming up with a diagnosis, and prescribing an approved patentable drug or recommending surgery or some other invasive procedure. Activity outside this paradigm is often ignored, ridiculed, or attacked. There is not much emphasis on finding underlying causes such as environmental toxins, suboptimal diets, poor lifestyle, and similar factors in interaction with the genotype of the person. Epigenetics is largely ignored. Today, unlike the issue in the Semmelweis case more that 100 years ago, money is a major factor. The entire health-care industry is supported by the wealthy pharmaceutical industry, as is virtually all medical school education and postgraduate training. The industry's many lobbyists have profound effect on legislation in the US and largely control the media. Legislators have passed legislation to exempt pharmaceutical companies from harm caused by vaccines while getting legislators and the various governmental agencies to mandate vaccines. This is true, even though evidence continues to mount that current vaccine schedules are causing undue harm such as an increased rate of autism, hyperactivity, asthma, allergies, and even cancer in children. Public authorities with reputations to uphold and a continuous supply of money from pharmaceutical companies continue to deny any link and use their authority to denigrate and ridicule those who criticize the medical paradigm.
The entire FDA system of drug approval is designed to keep this system of patentable drugs flowing while downplaying the approaches described in this article. The system is further reinforced by the FTC that attacks anything that sounds like a health claim, if it is not backed by the FDA, even though it may be true.
In spite of all of this, the public is becoming wiser largely as a result of more information available via the Internet and more and more people's taking charge of their health, making their own decisions rather than just relying on their doctors or the industry-supported media. It is essential that forward-thinking physicians begin to understand the new paradigm relating to health care and begin to incorporate this new knowledge into their care of patients in order for us to make progress in health care and, indeed, the survival of our planet.
Part 1, Part 2 and Part 3 are also online.

Notes
93. Beljanski M. The Regulation of DNA Replication & Transcription. New York: EVI Liberty Corp.; 2003.
94. Beljanski M. The regulation of DNA replication & transcription: the role of trigger molecules in normal and malignant gene expression. Exp Biol Med. 1983;8:1–190.
95. Nordau CG, Beljanski MS. Beljanski: A Pioneer in Biomedicine: Concepts, Theories and Applications. EVI Liberty; 2003.
96. Beljanski M. Oncotest: a DNA assay system for the screening of carcinogenic substances, IRCS Med Sci. 1979;7:476.
97. Beljanski M, Goff L LE, Beljanski MS. Differential susceptibility of cancer and normal DNA templates allows the detection of carcinogens and anticancer drugs. Third NCI-EORTC Symp. On new drugs in cancer therapy. Institut Bordet, Brussells; 1981.
98. Beljanski M, Beljanski MS. Three alkaloids as selective destroyers of cancer cells in mice. Synergy with classic anticancer drugs. Oncology. 1986;43:198–203.
99. Beljanski M, Crochet S. The anti-cancer agent PB100 concentrates in the nucleus and nucleoli of human glioblastoma cells but does not enter normal astrocytes. Int J Oncol. 1995;7:81–85.
100.Beljanski M, Crochet S, Beljanski MS. PB-100: a potent and selective inhibitor of human BCNU resistant glioblastoma cell multiplication. Anticancer Res. 1993;13:2301–2308.
101.Beljanski M, Crochet S. The selective anticancer agent PB-100 inhibits interleukin-6 induced enhancement of glioblastoma cell proliferation of human glioblastoma cell proliferation in vitro. Int J Oncol. 1994;5:873–879.
102.Beljanski M, Crochet S. Selective inhibitor (PB-100) of human glioblastoma cell multiplication. J Neurooncol. 1994;21:62.
103.Beljanski M, Crochet S. Mitogenic effect of several inter leukins, neuromediators and hormones on human glioblastoma cells, and its inhibition by the selective anticancer agent PB-100. Deutsche Z Onkol. 1996;28:14–20.
104.Beljanski M, Crochet S. The selective anticancer agents PB-100 and BG-8 are active against human melanoma cells, but so not affect non malignant fibroblasts. Int J Oncol. 1996;8:1143–1148.
105.Bemis DL, Capodice JL, Desai M, et al. b-Carboline alkaloid–enriched extract from the Amazonian rain forest tree pao pereira suppresses prostate cancer cells. J Soc Integr Oncol. 2009;7:59–65.
106.Beljanski M, Plawecki M. Particular RNA fragments as promoter of leucocyte and platelet formation in rabbits. Exp Cell Biol. 1979;47:218–225.
107.Beljanski M, Plawecki M, Bourgare P, et al. Short chain RNA fragments as promoters of leucocyte and platelet genesis in animals depleted by anti-cancer drug. In: Niu MC, Chuang HH, eds. The Role of RNA in Development and Reproduction. Sec. Int. Symposium. April 25-30, 1980. Beijing: Science Press; Van Nostrand Reinhold Company. pp. 79-113.
108.Plawecki M, Beljanski M. Comparative study of Escherichia coli endotoxin, hydrocortisone and Beljanski Leucocyte Restorers (BLR) activity in cyclophosphamide-treated rabbits. Proc Soc Exp Biol Med. 1981;168:408–413.
109.Beljanski M, Plawecki M, Bourgare MS, et al. Leucocyte recovery with short-chain RNA fragments in cyclophosphamide treated rabbits. Cancer Treat Rep. 1983;67:611–619.
110.Donadio, D, Lorho R, Causse JE, et al. RNA fragment (RLB) and tolerance of cytostatic treatments in hematology: a preliminary study about two non-Hodgkin's malignant lymphoma cases. Deutsche Z Onkol. 1991:23:33–35.
111.Beljanski M. Radioprotection of irradiated mice – mechanisms and synergistic action of WR-2721 and R.L.B. Deutsche Z Onkol. 1991;23(6):155–159.
112.Walker M. Personal communication.
113.Cuasse JE, Nawrocki T, Beljanski M. Human skin fibrosis RNase search for a biological inhibitor-regulator. Deutsche Z Onkol. 1994;26(5):137–139.
114.Burzynski SR. The present state of antineoplaston research (1). Integr Cancer Ther. 2004;3(1):47–58.
115.Elias TD. The Burzynski Breakthrough: The Most Promising Cancer Treatment … and the Government's Campaign to Squelch It. Rev. ed. Nevada City, CA: Lexicos; 2001.
116.Burzynski: The Movie. See www.burzynskimovie.com.
117.Fujii T, Nakamura AM, Yokoyama G, et al. Antineoplaston induces G1 arrest by PKCa and MAPK pathway in SKBT-3 breast cancer cells. Oncol Rep. 2005;14:489–494.
118.Matono K, Ogata Y, Tsuda H, et al. Effects of antineoplaston AS2-1 against post-operative lung metastases in orthtopically implanted colon cancer in nude rat. Oncol Rep. 2005;13:389–395.
119.Tsuda H, Sata M, Kumabe T, et al. The preventive effect of antineoplaston AS2-1 on HCC recurrence. Oncol Rep. 2002;10:391–397.
120.Tsuda H, Sata M, Ijuuin H, et al. A novel strategy for remission induction and maintenance in cancer therapy. Oncol Rep. 2002;9:65–68.
This is an expanded version of a lecture presented at the International Society of Integrative Medicine meeting in Tokyo Japan on July 19, 2009. The lecture was partially supported by Maitake Mushrooms Inc. (In the US, the company is now called Mushroom Wisdom Inc. The full article was originally published in the International Journal of Integrative Medicine July 2010;2(1) and is republished in parts in a slightly modified form with permission.

|



![]()
![]()
![]()







 Perhaps the most famous patient who benefitted from the Beljanski products was the late former French President Francois Mitterrand (
Perhaps the most famous patient who benefitted from the Beljanski products was the late former French President Francois Mitterrand (