Natural
Therapies for the Prostate Gland:
A Scientific Review of Nutrient and Herbal Combinations
that Promote
Prostate Health
The prostate is a walnut-sized gland that sits at
the base of the male bladder, surrounding the prostatic urethra.
As part of the reproductive
system, this gland secretes seminal fluid, energizes sperm and provides
for it a favorable alkaline medium. The prostate is subject to several
disease states including infection, hypertrophy and cancer, causing
pain, urinary frequency and urgency, inability to urinate, painful
urination and sexual dysfunction.
Benign prostatic hyperplasia (BPH), prostate cancer, and prostatitis
are its most common disorders. More than 50% of men in their sixties
and as many as 90% of men in their seventies and eighties are diagnosed
with BPH, resulting in 375,000 hospital stays each year in the United
States alone. Prostate cancer is second only to cancer of the lung
as the leading cause of cancer death among men. Each year, more than
100,000 new cases of prostate cancer occur in the US, and more than
30,000 deaths are caused annually by this disease.1 The good news is
that over the past 20 years, the survival rate for prostate cancer
has increased from 67% to 97%.2
Unfortunately, benign prostatic hyperplasia (BPH) and prostate cancer
do not have a known etiology, making prevention difficult and treatment
less than ideal. Standard lines of treatment for these conditions,
including pharmacological preparations, surgical and non-surgical procedures,
hold significant risks. Additionally, as new theories about the functioning
of the prostate gland develop, scientists are questioning the effectiveness
and the rationale behind the continued use of these treatment modalities.
A close review of the medical literature verifies the safety and efficacy
of nutritional and botanical therapeutic agents such as Serenoa repens
(saw palmetto), Urtica dioica (stinging nettle root), Prunus africanum
(pygeum), essential fatty acids, selenium, green tea extract, tomatoes
and zinc to promote and maintain prostate health and alleviate urologic
symptoms. In addition, select amino acids, in relatively physiologic
doses, exhibit promising clinical results for the prostate patient.
These natural ingredients effect a positive change in prostate health
without the associated risk of adverse reactions. Following is a review
of the most common prostate conditions and available treatment options,
including a comprehensive evaluation of herbal and nutrient therapies
for the prostate gland, scientific validity, positive patient outcomes,
and incidences of negative side effects.
Benign Prostatic Hyperplasia
(BPH; Prostate Enlargement; Lower Urinary
Tract Symptoms (LUTS))
As a man ages, his prostate gland typically begins to enlarge. The
medical community has no well-defined reason for this phenomenon, which
is identified as benign prostatic hyperplasia (BPH), benign prostatic
hypertrophy, or lower urinary tract symptoms (LUTS). This condition
does not generally cause symptoms until a man reaches the age of 50,
with the majority of associated problems peaking by age 70. BPH causes
the prostate gland to constrict the urethra, making micturition increasingly
difficult. As the bladder is forced to contract against increasing
resistance, the bladder muscle (the detrusor) becomes hypertrophied
and irritable. As the condition progresses, the bladder becomes unable
to empty completely, and finally to empty at all. Residual urine in
the bladder sets the stage for serious bladder infections and kidney
malfunction.
Symptoms of BPH include:
· Reduced caliber and force of urine stream
· Frequent urination (particularly in the evening)
· Urinary urgency
· Leaking or dribbling
· Urinary retention (inability to pass urine)
Occasionally, a man may not realize that he has a urinary obstruction
until he finds himself unable to urinate at all. Physicians refer to
this type of episode as acute urinary retention, which can be triggered
by a wide variety of drugs with anticholinergic side effects, both
prescription and over-the-counter, such as anti-depressants, cold and
allergy medications. These medications can tighten the bladder outlet,
resulting in urinary retention. Alcohol, cold temperatures or a long
period of immobility can also cause urinary retention when there is
a partial obstruction present. Severe BPH can have serious long-term
health effects such as urinary tract infections, bladder or kidney
damage, bladder stones, painful intercourse, and incontinence.
Conventional Therapies for BPH
Finasteride (Proscar®), terazosin (Hytrin®), doxazosin (Cardura®),
and tamsulosin (Flomac®) are medications approved by the FDA for
the treatment of BPH. These pharmaceuticals aim at blocking DHT production
or relaxing the prostatic urethra sphincter. Specifically, finasteride
(Proscar®) inhibits the production of dihydrotestosterone (DHT),
while terazosin (Hytrin®), doxazosin (Cardura®), and tamsulosin
(Flomac®) force smooth muscle relaxation of the prostate and bladder
neck. Nonsurgical treatments include transurethral microwave procedures
(TUMT) and transurethral needle ablation (TUNA). Surgical treatments
include open, laser and transurethral (TURP) surgery. These treatments
remove or destroy sections of the prostate gland. Botanical and nutritional
therapies for BPH focus on decreasing the size of the prostate gland,
preventing abnormal cell growth, and supplying the gland with nutrients
to decrease the risk of infection, while maintaining hormonal balance
and preserving the complete gland.
A Closer Look at Finasteride (Proscar)
Finasteride (Proscar®), a popular
prescription for BPH, functions by interfering with the action of 5
alpha-reductase, the enzyme that
converts testosterone into DHT in the prostate gland. DHT is an androgen
that stimulates the synthesis of specific proteins and causes prostate
cells to proliferate. The theory behind finasteride is that the body
is producing too much testosterone and ultimately DHT, which is causing
abnormal cell proliferation. By blocking the action of 5 alpha-reductase,
the prostate gland should shrink.
Merck sponsored a double-blind placebo controlled study using finasteride
(Proscar®) that demonstrated significant reductions in DHT,3 improvements
in urinary function,4 and decreases in prostate volume5 in
men with BPH. A bias may be present considering the sponsor of this
research
although there are more pressing issues regarding the use of finasteride.
First, finasteride (Proscar®) only decreases the size of the prostate
gland by 18% even though it reduces DHT levels by a surprising 80%.
Further, only 37% of men using the drug for an entire year experience
any symptom improvement. Sexual side effects of finasteride include
decreased libido, impotence, and ejaculatory disorders.6 Finasteride
(Proscar®) also increases a man's risk of developing prostate
cancer.7 While this drug is in fact lowering DHT levels,
it is clearly not promoting the health of the prostate gland. Moreover,
the theory
that DHT is the primary cause of abnormal cell growth is scientifically
invalid, since a dramatic decrease in DHT levels plays a minimal role
in preventing abnormal cell growth, and may even promote it. This theory
represents a narrow and disconnected view of prostate function. A man's
serum testosterone level falls as he ages. If testosterone causes BPH,
then why would a man's risk of developing BPH increase as his
levels of testosterone decrease? There is clearly more to this picture
than that which medical scientists currently accept as fact.
The answer to this dilemma might be the growing imbalance between estrogen
and testosterone levels in aging men. While estrogen levels remain
relatively unchanged, testosterone or androgen levels fall dramatically
as a man ages. However, in the stroma of the prostate (the area of
tissue where BPH is thought to develop), DHT levels remain constant,
while estrogen levels dramatically increase. The stroma produces additional
estrogen (specifically estradiol) from testosterone in a process known
as aromatization. Human sex hormone-binding globulin (SHBG) also affects
the estrogen/testosterone ratio, since levels of SHBG increase with
age. Normally, androgens like testosterone have an affinity for SHBG
and cause cell proliferation. Medical researchers are discovering,
however, that estrogen binds to SHBG as well, causing abnormal cell
proliferation in the prostate. This information becomes particularly
pertinent when viewed in light of the mechanisms of action of saw palmetto,
nettle root and pygeum (discussed in detail in subsequent sections
of this paper), and their clinical efficacy in the prevention and treatment
of BPH and cancer.8
A Closer Look at Prostate Surgery
Most physicians recommend removal of the enlarged part of the prostate
as the best long-term solution for patients with BPH.9 After any
surgery for BPH, a Foley catheter is temporarily inserted through
the penis to drain urine from the bladder into a collection bag while
the surgical site is healing. This catheter frequently causes infection
and recurring painful bladder spasms. Sexual function is affected
in up to 30% of surgical cases, including retrograde ejaculation
(which causes sterility). Furthermore, surgery for BPH does not prevent
future prostate problems since a portion of the gland remains. Finally,
scar tissue from the surgery typically forms in the urethra and causes
narrowing (which is what the surgery is supposed to relieve). Patients
with severe BPH who have already failed botanical and nutrient combinations
and drug therapies may require surgical intervention to restore urinary
flow. In order to prevent surgical intervention, it is essential
to utilize noninvasive therapies at the onset of BPH symptoms or
before. Medical professionals use pharmaceutical agents and surgical
treatments as a standard approach to managing patients with moderate
BPH. The risks of such therapies can outweigh their benefits. However,
other scientifically valid therapies, including key nutrient and
herbal combinations, are at least as effective clinically, without
the associated risks.
Prostate Cancer
Cancer of the prostate gland is the most commonly diagnosed non-dermatological
cancer among men. It is a leading cause of cancer death among American
men, second only to lung cancer. In the United States, more than
30,000 men die each year from prostate cancer. One in five men will
develop prostate cancer in his lifetime, and three percent of those
will die from it.
The condition occurs when normal prostate cells turn cancerous (malignant)
and divide at an unreasonable rate. It most often develops in the region
of the prostate closest to the rectum. African-American men, men aged
65 and older, and men who have a first-degree relative with prostate
cancer are at an increased risk. Black Americans have the highest incidence
in the world. Interestingly, there is a direct correlation between
intake of animal fat and the risk of getting prostate cancer. The National
Cancer Institute established that total consumption of animal fat directly
relates to the risk of advanced prostate cancer. Red meat has the strongest
positive association with advanced prostate cancer.10 It is critical
to note that the association is only with saturated animal fat. In
fact, essential fatty acids (EFAs), given in the correct ratios, may
prove to be a key in preventing prostate cancer, and even promoting
the health of the gland.
Symptoms
There are essentially no warning signs for early prostate cancer. As
the cancer advances, some men may experience symptoms similar to
those associated with BPH (urinary frequency, urgency, pain with
urination, etc.) or a urinary tract infection.
Early Detection and Treatment
There is no scientific evidence that provides a definite link between
early detection and even treatment of prostate cancer and the incidence
of deaths caused by this disease.11 Further, if a health
professional diagnoses a man with early stage prostate cancer, it
is highly likely
that he will then be required to undergo invasive evaluation. If
this evaluation results in cancer detection, then standard treatments
are generally employed. Standard treatments such as surgery, radiation
therapy, hormonal therapy and occasionally chemotherapy have serious
side effects including incontinence (lack of bladder control), impotence
(inability to have an erection), and even death. One treatment
option for men with early-stage prostate cancer is "watchful
waiting." Since many prostate cancers are small and grow slowly,
physicians may opt to shun treatment altogether (watchful waiting),
which is an accepted approach to managing the condition. A benefit
of this method is that it avoids any potential side effects that
may occur with other forms of treatment. Men choosing this method
of treatment (or lack thereof) often turn to nutrient and botanical
therapies as a means of safely treating their cancer. Interestingly,
a study completed at the University of Pennsylvania Hospital12 found
that one third of all prostate cancer patients supplement medical
treatment with complementary therapies.
Prostatitis
Acute bacterial prostatitis is an acute infection of the prostate gland.
Bacteria usually migrate to the prostate from the urinary system.
As the infection advances, the gland begins to swell, causing sudden
symptoms of painful, urgent and frequent urination. Other symptoms
include fever and chills, and pain over the bladder, lower back and
between the testicles and rectum (the perineal area). The standard
medical treatment for acute prostatitis is usually intravenous antibiotics
until the fever breaks, followed by 4-6 weeks (or longer) of oral
antibiotic therapy. It is critical that one seeks medical attention
promptly if he experiences fever-associated pain in the areas described
above.
Chronic bacterial prostatitis is usually symptomatic, causing vague,
persistent, low back and perineal pain, urinary urgency and frequency,
and painful urination. Standard medical treatment is oral antibiotic
therapy for 2 to 3 months. Patients can also experience prostatitis
that is not the result of a bacterial invasion. This condition is referred
to as chronic non-bacterial or non-infectious prostatitis. Chronic
non-bacterial prostatitis is more common than bacterial prostatitis,
and has the same symptom picture. Having no known cause, healthcare
professionals find it difficult to treat. It does not respond to antimicrobial
agents. Serenoa repens (saw palmetto), Urtica dioica (stinging nettle
root), Prunus africanum (pygeum), zinc, selenium, green tea extract,
tomatoes, amino acid therapy, and essential fatty acid therapy are
effective and safe treatment options for nonbacterial, non-infectious
prostatitis.

Botanical and Nutritional Therapeutic Options for Prostate Conditions
Serenoa
repens
(Saw Palmetto)
Description and Medical History
Saw
palmetto is native to North America. It is a member of the fan palm
family and has characteristic
sharp
edges that can literally "saw" through clothing, hence
its common name, saw- palm-etto. The plant produces a one-seeded
dark brown-to-black berry that is harvested and used in the preparation
of phytomedicines. Researchers now focus on the lipophilic medicinal
extracts of these berries, although one should not ignore its historic
use as a whole berry preparation. Traditionally used for the treatment
of prostate conditions, saw palmetto was appropriately referred to
as the "old man's friend." Touted for its effectiveness
in reducing prostate inflammation, alleviating chronic urinary tract
infections, and increasing sperm count, this herb has demonstrated
the added benefit of increasing sex drive in men.13,14
Known Medicinal Constituents
· Lipoid constituents: Phytosterols,
particularly beta-sitosterol, tri, di, and monoglycerides, and free
fatty acids
· Flavonoids and polysaccharides
Indications for Use (specific to prostate health)
· Mild to moderate benign prostatic
hyperplasia14-16
· Non-infectious (non-bacterial) prostatitis17
· Prostate cancer18,19
Possible Mechanisms of Action
· Has alpha-adrenoreceptor and
calcium blocking activities (antagonists) that function to relieve
urinary urgency by reducing the smooth muscle
contractions of the bladder sphincter20
· Inhibits the binding of DHT to androgen receptor sites in the cytosolic
component of prostate cells21,22
· Inhibits prolactin-induced prostate growth23
· Inihibits the binding of growth stimulating estrogen to receptor sites
on the prostate cells and reduces the number of estrogen and androgen
receptors in the nuclear fraction of the prostate cell24
· Exhibits mild 5-alpha reductase inhibition activity in the prostate
gland25,26
· Inhibits the arachadonic acid cascade via the inhibition of cyclooxygenase
and 5-lipoxygenase in the prostate27
Double-Blind Clinical Trials with Saw Palmetto in BPH
Wilt et al.28 conducted a systematic review and quantitative meta-analysis
of the existing evidence regarding the therapeutic efficacy and safety
of saw palmetto plant extracts in men with mild to moderate benign
prostatic hyperplasia (BPH). The researchers evaluated 18 randomized
controlled trials that included 2,939 men with a mean age of 65 years.
Sixteen of these studies (89%) were double-blinded and placebo controlled,
and the mean duration of the studies was 63 days. The researchers
measured the efficacy of saw palmetto extracts in affecting urologic
symptoms, urine flow, residual urine volume, prostate size and nocturia.
The study participants (that were treated with saw palmetto) and
their physicians reported significant improvements in BPH symptoms
compared to the placebo group. The men had decreased urinary tract
symptom scores overall, fewer episodes of nocturia (-0.76 times per
night), and an increase of peak urine flow rates (1.93 mL/s [95%
CI, 0.72-3.14]). All trials in this meta-analysis produced similar
improvement in urinary tract symptoms and urinary flow rates when
measured against finasteride, and were associated with significantly
fewer adverse side effects.
Table 1.1 highlights seven key placebo-controlled clinical trials with
a liposterolic extract of saw palmetto, all of which were a part of
the recent systematic review by Wilt et al.28 In six out of seven of
the studies, urinary frequency was dramatically decreased in the saw
palmetto treatment group as compared to the placebo group. The saw
palmetto group experienced a significant improvement in peak flow rates
(26-50%) compared to the placebo group (2-35%). While symptom score
evaluations are a valuable means of analyzing outcomes, the researchers
did not routinely complete these evaluations since many of the studies
were done before this methodology was widely accepted.29
Table 1.1 Double Blind Placebo-Controlled Clinical
Trials of Saw Palmetto Extract in BPH Patients
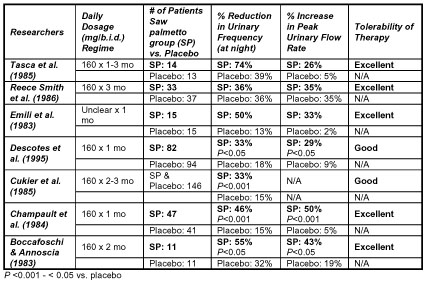
Use this
link to view a larger Table 1.1, as a .pdf. Finasteride vs. Saw Palmetto Extract
Carraro et al.30 completed the largest international comparative trial
for the treatment of BPH. This double-blind study compared the therapeutic
effects of finasteride versus saw palmetto extract in 1,098 patients
with moderate BPH. The most critical observation of this comparative
trial was that saw palmetto extract does not have a significant effect
on serum PSA levels or prostate volume. With this knowledge, physicians
do not need to be concerned that saw palmetto will affect PSA levels
and mask prostate cancer. This clinical evidence also makes it clear
that the primary action of saw palmetto is not its ability to inhibit
5-alpha reductase, since PSA levels remained relatively constant.
Table 1.2 further highlights the results of this study.
Table 1.2 Double-Blind Trial Comparing the Therapeutic Effects of
Saw Palmetto Extract with Finasteride in Patients with Moderate BPH30
| Treatment |
Saw Palmetto Extract |
Finasteride |
| Daily Dosage |
320 mg |
5 mg |
| Quality of Life |
38% improvement |
41% improvement |
| Peak Flow Rate |
25% |
30% p<0.05 |
| Prostate Volume |
-6% |
-18% p<0/001 |
| PSA |
No change |
-41% p<0/001 |
| IPSS |
-37% |
-39% |
Patients reported more sexual dysfunction with
finasteride and reported a significantly higher number of incidences
of impotence
and decreased
libido p<0.001 on sexual function score
IPSS= International Prostate Symptom Score
PSA= prostate-specific antigen

Overview
Clinical Effects of Saw Palmetto (Serenoa repens)
· Decreases urinary urgency
· Decreases perineal pain
· Decreases nocturnal (nighttime) frequency
· Increases urinary flow rate
· Prevents infections due to residual urine
· Reduces residual urine
· Improves quality of life for BPH patients
Contraindications
Pregnancy and lactation: it is possible that Saw palmetto may be unsafe
for pregnant and lactating women due to its antiandrogen and estrogenic
activity.31
Side Effects
Tolerability is generally excellent for Saw palmetto although it can
in rare instances cause headache or mild gastrointestinal disturbances.30
Possible Interactions with Drugs
May interfere with oral contraceptives and hormone therapy due to it
antiestrogen effect.24
Possible Interactions with Herbs and other Dietary Supplements
None known
Possible Interactions with Lab Tests
None: Saw palmetto does not have a significant effect on serum PSA
levels.30
Recommended Dosage
Lipophilic Extract: 160 mg b.i.d., according to research studies referenced
in this review (may be reduced if combining with dried berries)
Dried berry: .5-1 gram t.i.d.

Urtica dioica
(Stinging Nettle Root)
Description
and Medical History
Nettle is a weed that has a particular affinity
for nitrate-rich soil, found in most temperate regions of
the world. Its root and rhizome (the parts used today for the
treatment of prostate conditions) are long and yellow-brown in color.
According
to the Eclectics, nettle root (including the rhizome) relieved
bronchial and asthmatic trouble. Healers also used nettle as a diuretic
and
astringent, and as a treatment for joint ailments.32 Its high
lignan content made it an attractive fiber source before the introduction
of flax seeds.
As mentioned earlier in this review, estrogen and androgens such
as testosterone bind to SHBG and cause cell proliferation. As
a man ages,
his SHBG levels increase, making him more susceptible to abnormal
cell proliferation. Today, researchers are finding the high lignan
content
of nettle to be responsible for the root's ability to bind to
SHBG (sex hormone binding globulin), a key to its potential use
as a prostate cancer remedy.33,34 This strong affinity for SHBG
limits
the amount of testosterone and estrogen that can bind to it and
influence cell proliferation.
Known Medicinal Constituents
· Scopoletin (a coumarin)
· Lignans (Urtica dioica agglutinin [UDA], (-)-secoisolariciresinol)
· Sterols and steryl glycosides (including sitosterols)
Indications for Use (specific to prostate health)
· Mild to moderate benign prostatic
hyperplasia34,35
· Non-infectious (non-bacterial) prostatitis34,36,37
· Prostate cancer33
Possible Mechanisms of Action
· Inhibition of sodium-potassium
ATPase from BPH tissue, thereby decreasing androgen response8
· Reduction of human sex hormone-binding globulin (SHBG) activity due
to lignans found in nettle root 34,38
· Antagonism of the pathway by which SHBG leads to the induction of androgen-responsive
genes39
· Inhibition of human leukocyte elastase (HLE), a marker in prostatitis
and associated lower urinary tract infections (LUTI)37
· Weak inhibition of DHT binding to cytosolic androgen receptors in the
prostate39
· Inhibition of aromatase, thereby inhibiting the conversion of androgens
to estrogens38
Placebo Controlled Clinical Trials Involving Stinging Nettle Root
in BPH patients
The popular use of stinging nettle root extract in Germany encouraged
the onset of numerous clinical trials. Collectively, these trials demonstrated
the benefits of using nettle root for the treatment of BPH. In a 1996
placebo controlled trial, Engelmann et al.40 successfully demonstrated
that nettle root extract was superior to placebo in terms of the International
Prostate Symptom Score (IPSS). The study included 41 subjects with
mild BPH, observed for a period of 3 months. In an older placebo controlled
trial (1987),41 79 patients with BPH were given either nettle root
extract or placebo for as long as two months. The dosage was 600 mg
per day for the duration of the study. Nettle root proved to be better
than the placebo in all parameters that were measured including urinary
flow, residual urine and urinary volume. In 1985, Vontobel et al.42 conducted
a placebo controlled clinical trial that demonstrated a significant
improvement in urinary flow and micturition volume, and an impressive
decrease in SHBG (p<0.0005). The study included 50 patients that
were prescribed 600 mg of nettle root extract daily for approximately
two months.
Placebo Controlled Clinical Trials of Stinging Nettle Root/Saw Palmetto
Combination Therapies
Metzker et al.43 completed a double blind placebo controlled clinical
trial involving 40 BPH patients that took either a placebo treatment
or a mixture of nettle root and Saw palmetto extracts for 6 months.
The experimental group showed a dramatic improvement in IPSS (International
Prostate Symptom Score) as compared to the placebo group. Additionally,
urinary flow rates improved significantly in the experimental group
compared to the placebo group. Details of this study are highlighted
in Table 2.1. The 6-month double blind treatment was followed by an
open-label extension. Interestingly, the placebo group demonstrated
a marked improvement in urinary flow rates from 15.50 to 17.5 ml/sec.
However, the group receiving the saw palmetto/ nettle extract demonstrated
a further statistically significant increase in urinary flow rate with
continued therapy for one year.
Table 2.1. Saw Palmetto/Nettle Root Trial43
| |
320mg Saw Palmetto Extract + 240 mg Nettle Root Extract for 6
months |
Placebo for 6 months |
| IPSS |
18.3 - 11.1 (40% decrease in symptoms) p<0.05 |
19.0-17.67 (7% decrease in symptoms) p<0.05 |
| Urinary Flow Rates |
Improved from 14.65 - 17.95 ml/sec (23% improvement) |
Improved from 15.05 - 15.50 ml/sec (4% improvement) |
IPSS = International Prostate Symptom Score
Sokeland et al.44 completed another comparison trial with finasteride
and a saw palmetto/nettle root extract.
Table 2.2 Saw Palmetto/Nettle Root vs. Finasteride
| |
Daily Dosage |
Duration |
IPSS % Change |
| Finasteride Group |
5 mg |
12 months |
47% Improvement - 5.6 point change |
| Saw Palmetto/ Nettle Root Group |
160 mg saw palmetto liposterolic extract/120 mg nettle root extract
b.i.d |
12 months |
42% Improvement - 4.8 point change |
IPSS = International Prostate Symptom Score
This randomized, double-blind, multicenter clinical trial involved
543 patients suffering from BPH stages I and II. Both therapies proved
to be similar in effectiveness in all areas including: IPSS scores,
urination times, and urinary flow rate. The major difference was in
tolerability. Patients tolerated herbal therapy better than finasteride.
The finasteride group reported more adverse events including diminished
ejaculation volume, erectile dysfunction and headaches. Table 2.2 highlights
dosages, duration and IPSS scores for this study.

Overview
Clinical
Effects of Stinging Nettle Root (Urtica dioica)
· Decreases urinary urgency
· Decreases nocturnal (nighttime) frequency
· Encourages hormonal balance
· Increases urinary flow rate
· Prevents infections due to residual urine
· Reduces residual urine
· Reduces risk of abnormal prostate cell proliferation
· Improves quality of life for BPH patients
Contraindications and Warnings
Pregnancy and lactation: insufficient reliable information. Avoid using
unless under the strict supervision of a qualified healthcare practitioner.
Benign prostatic hyperplasia (BPH) therapies and prostate cancer
therapies should be monitored by a qualified healthcare practitioner.
Side Effects
Tolerability is generally excellent for nettle root.45
Possible Interactions with Drugs: None known
Possible Interactions with Herbs and other Dietary
Supplements: None
known
Possible Interactions with Lab Tests: None known
Recommended Dosage
Dried extract (5:1) 600-1200 mg/day, according to research studies
referenced in this review
Use stinging nettle root for prostate symptoms, not to be confused
with white dead nettle, which is primarily used for inflammation of
the mucous membranes.

Pygeum africanum
(Pygeum)
Description and Medical History
Pygeum, sometimes
referred to as African Plum Tree, is a large evergreen tree that
grows in the higher plateaus of southern
Africa. The Eclectics and other healers traditionally collected the bark
of the tree, ground it into a powder, and prescribed it as a tea for genito-urinary
conditions. It was also used as an aphrodisiac, and as a remedy for "madness."46
Medical researchers now consider pygeum bark to be a scientifically valid
and clinically effective remedy for functional symptoms of benign prostatic
hyperplasia (BPH) that include nocturia, dysuria, mictritional disorders,
and bladder fullness.44,47-52
Known Medicinal Constituents
· Phytosterols (including beta-sitosterol,
beta-sitosterone and campesterol)
· Ferulic esters of long chain fatty acids
· Pentacyclic triterpenes (including oleanolic, crataegolic and ursolic
acid)
Indications for Use (specific to prostate health)
· Mild to moderate benign prostatic
hyperplasia.44,53-58
· Non-infectious (non-bacterial) prostatitis32,51
· Prostate cancer50
Possible Mechanisms of Action
· Beta-sitosterols found in
Pygeum inhibit prostaglandin synthesis, thereby reducing the abnormally
high levels of prostaglandins normally found
in BPH patients.36
· Pygeum prevents bladder contractile dysfunction, thereby reducing risk
of prostatitis and/or BPH complications.49
· Pygeum is a potent inhibitor of prostatic fibroblast proliferation
via its ability to inhibit protein kinase C activation. Rapidly growing
benign and malignant cells require protein kinase C activity50
· Pygeum antagonizes 5-lipoxygenase metabolite production, thereby decreasing
bladder hyperreactivity and prostate inflammation.27
· Ferulic acid esters reduce prostatic cholesterol levels, thereby limiting
synthesis of testosterone.32
· Triterpens, including oleanolic, crataegolic and ursolic acid decrease
inflammation in prostate tissue.
· Pygeum competes with androgen precursors.32
Placebo-Controlled Clinical Trials Involving Pygeum Bark and BPH patients
Pygeum bark extract is a popular treatment in Europe, particularly
in central and Eastern Europe. Breza et al.48 conducted a multicenter
trial in central Europe in 1998 to determine the efficacy and safety
of using Pygeum africanum extract in the treatment of BPH. The researchers
evenly distributed 85 patients with mild to moderate BPH among three
centers. IPSS scores decreased by 40% and quality of life scores
by 32%. There was also a statistically significant reduction in nocturnal
frequency (32%) although urinary flow parameters remained unchanged.
The researchers reported a particularly satisfactory safety profile.
An interesting aspect of this study was the researchers' decision
to follow the patients after the initial treatment for an additional
month with no treatment. The results of the treatment remained positive
even after it was stopped, indicating a persistent therapeutic activity.
Table 3.1 highlights the results of this study.
Table 3.1 Pygeum africanum extract in the treatment of BPH48
| |
TWO MONTH PERIOD OF TREATMENT WITH 50 MG, b.i.d. |
ONE MONTH FOLLOW-UP WITH NO TREATMENT |
| IPSS |
40% mean improvement |
Maintained clinical improvement |
QOL
(Quality of Life) |
31% mean improvement |
Maintained clinical improvement |
| Nocturnal Frequency |
32% mean reduction |
Maintained clinical improvement |
| Tolerability of Treatment |
Excellent |
N/A |
Subjects: 85 men aged 50-75 with mild to moderate BPH
IPSS=International Prostate Symptom Score
Chatelain et al.47 completed a more recent study that compared the
efficacy and safety of Pygeum africanum extract at different dosages.
This randomized, double blind study included 209 patients in its first
phase (2 months long using either 50 mg twice daily or 100 mg once
daily) and 174 patients in its second phase (10-month, open phase using
100 mg once daily). IPSS, quality of life (QOL) and maximum flow rate
(Qmax) improved significantly in all patients, and both treatments
had similar efficacy. The researchers reported a satisfactory safety
profile after using 100 mg per day for a period of 12 months. Table
3.2 summarizes the significant results of this study.
Table 3.2 Comparison of Efficacy and Safety of Pygeum africanum Extract
Administered as 50 mg bid and 100 mg qd47
| |
Group A: 50 mg Twice Daily |
Group B: 100 mg Once Daily |
| IPSS (2 month comparative phase) |
38% mean improvement |
35% mean improvement |
| QOL (2 month comparative phase) |
28% mean improvement |
28% mean improvement |
| Qmax (2 month comparative phase) |
16% increase |
19% increase |
| Tolerability (2 month comparative phase) |
Excellent |
Excellent |
| 10 Month Open Phase |
100 mg Once Daily |
| IPSS |
46% Improvement |
| Qmax |
15% increase |
| Tolerability |
Excellent |

Overview
These clinical trials coupled with the results of earlier
in vitro trials clearly show that Pygeum africanum bark extract:
· Decreases nocturnal frequency
· Decreases urinary urgency
· Improves quality of life for BPH patients
· Inhibits prostate cell proliferation
· Reduces prostate size
· Reduces residual urine
· Does not affect serum hormone levels
· Is very well tolerated
Contraindications
Pregnancy and lactation: insufficient reliable information. Avoid using
unless under the strict supervision of a qualified healthcare practitioner.
Side Effects
Tolerability is generally excellent for Pygeum africanum bark47,48
although some researchers speculate that it may, in rare instances,
cause nausea and mild abdominal pain.36
Possible Interactions with Drugs: None known
Possible Interactions with Herbs and other Dietary
Supplements: None
known
Recommended Dosage: 100-200 mg lipophilic extract (12-14%) per day
in 6-8 week cycles, according to research studies referenced in this
review
*It is advisable to be mindful of the source for pygeum used since
over harvesting may be threatening the species.46

Green Tea
The Prostate Cancer Research Institute reports on its web
page59 that green tea and a lipid-extract from it contain several
pharmacologically
active chemicals in the flavonol group of polyphenols known as
catechins. In addition to being anti-oxidants and free-radical scavengers,
these
compounds inhibit the expression of oncogene and the action of
three enzymes believed to play a role in oncogenesis. They have demonstrated
anti-cancer activity in laboratory mice carrying human prostate
and
breast cancers and mouse lung and skin cancers.
Two catechins found in green tea, epicatechin gallate (ECG), and
epigallocatechin-3 gallate (EGCG) are inhibitors of 5-alpha reductase
which may be effective
in the treatment of 5 alpha dihydrotestosterone-dependent abnormalities,
such as benign prostate hyperplasia and prostate cancer. It may be
possible that these catechins are regulating androgen action in the
prostate gland. As described earlier in this article, 5 alpha-reductase
is the enzyme that converts testosterone into DHT in the prostate
gland. DHT is an androgen that stimulates the synthesis of specific
proteins
and causes prostate cells to proliferate. The theory behind 5-alpha
reductase blocking agents is that the body is producing too much
testosterone and ultimately DHT, which is causing abnormal cell
proliferation. By
blocking the action of 5 alpha-reductase, the prostate gland should
shrink.60

Alanine, Glutamine, and Glycine
In 1958, Feinblatt and Gant61 completed
a controlled cross-over study using a combination of alanine, glutamine
and glycine to treat BPH
symptoms. The study included 40 men with confirmed BPH. After 3
months on the amino acid combination, the authors reported that delayed
micturition was either relieved or reduced by 70%, nighttime urinary
frequency was reduced by 95%, urinary urgency decreased by 81%
and
urinary frequency decreased by 73%. These preliminary results inspired
the work of Damrau in 1962. He conducted a controlled study that
included 45 cases of uncomplicated BPH in an experimental group
and 40 cases of uncomplicated BPH in a control group. The age, weight
and height of all subjects were closely matched. The experimental
group received the combination amino acid therapy daily for three
months. Table 4.1 reveals the results of this study.
Table 4.1 Amino Acid Therapy for BPH (Modified from Damrau F62)
| Symptoms |
Amino Acid Group
(% showing complete or partial relief of symptom) |
Placebo Group
(% showing complete or partial relief of symptom) |
Amino Acid Group
(% showing complete relief of symptom) |
Placebo Group
(% showing complete relief of symptom) |
| Nocturia |
56 |
15 |
15 |
5 |
| Urgency |
66 |
11 |
42 |
11 |
| Frequency |
43 |
15 |
33 |
0 |
| Delay in starting |
50 |
5 |
19 |
5 |
| Difficulty maintaining urination |
46 |
0 |
23 |
0 |
Damrau62confirmed the results of the original study conducted by
Feinblatt and Gant61 on the safety and efficacy of using a combination
of alanine,
glutamine, and glycine in the treatment of BPH. Damrau reported that
he observed no side effects in the experimental group.
In 1978 Cuervo et al.63 studied the effects of the amino acid combination
(alanine, glutamine, glycine) together with the herb, Prunus arborea.
They determined that the inclusion of the amino acids with the herb
led to a considerable reduction in the duration of treatment for BPH
patients resulting in significant financial savings. This study included
100 patients with prostatic adenoma, prostatitis, and BPH.
In Japan, this amino acid combination is available as Paraprost for
the treatment of BPH. Maekawa64 compared the effects of Paraprost with
another drug, Cernilton, on BPH. This multicenter double blind study
included 192 patients with BPH. The researchers determined that Paraprost
(the amino acid combination) was more than moderately effective in
46.3% of the patients studied. Yamaguchi et al. (1990) also compared
the effects of Paraprost with another drug (a selective alpha 1-adrenoreceptor
blocker) in a double-blind multi-institutional study. They observed
77 patients with BPH and determined that Paraprost significantly improved
obstructive and irritative symptoms, and reduced the incidence of residual
urine.
The combination of these three amino acids – alanine, glutamine
and glycine – offers favorable clinical results for the patient
with BPH. The therapy appears to enhance the positive effects of herbal
prostate medicines.

Zinc
The prostate gland has a unique characteristic of storing remarkably
high levels of zinc. In fact, healthy prostate tissue contains
a higher concentration of zinc than any other tissue in the human body.
It is no coincidence that the level of zinc in the prostate gland
declines dramatically in patients with BPH, and even more so in
patients
with prostate cancer. Zaicheick et al.65 studied 109 patients with
BPH (50 cases) and prostate cancer (109 cases). The researchers
used transrectal punch biopsy of prostate and radionuclide-induced
energy
dispersive X-ray fluorescent analysis to determine zinc concentrations
in prostate tissue. They showed that the zinc content in cancerous
prostate tissue was dramatically less than the concentration found
in healthy prostate tissue. Specifically, zinc content of the normal
prostate was 1018 micrograms/g dry tissue (M+/- 124) compared to
146 M+/-10 in the cancerous prostate tissue. Costello et al.66found
an interesting correlation between citrate metabolism in the prostate,
zinc accumulation, and prostate cancer. They determined that, in
addition to zinc, the prostate accumulates and secretes exceptional
levels of citrate from prostate epithelial cells. These cells will
not secrete normal amounts of citrate if the zinc level in prostatic
mitochondria drops. Specifically, the accumulation of zinc inhibits
an enzyme that causes citrate oxidation. Thus, in patients with
prostate cancer, as the level of zinc in the prostate drops, the threat
of
citrate oxidation increases. Perhaps the most interesting finding
was that prolactin and testosterone regulate the concentration
of zinc in the epithelial cells that produce citrate. Numerous medical
researchers have made the correlation between zinc levels and BPH
and cancer.65,67-71 Despite this correlation, there has been little
support for further investigation of the nutritional etiology of
BPH and prostate cancer. It is also important to note that preliminary
research suggests that over supplementation with zinc (over 100
mg
per day) for prolonged periods may increase risk of prostate carcinogenesis.
This is based on an epidemiological study72 where confounding variables
exist, including concurrent high dose supplement intake of other
ingredients such as calcium. Nevertheless, it does elucidate the
point that more is not necessarily better with dietary supplement
intake.

Tomatoes
Compared to other known carotenoids,
lycopene is considered by experts to be one of the most potent antioxidants,
with its
unsurpassed singlet-oxygen
quenching capacity. An impressive number of clinical trials and
epidemiological studies demonstrate the statistically significant association
between
consumption of lycopene in foods, such as tomatoes and prostate
health. However, most of the investigations are plagued by the uncertainties
common to dietary therapeutics – the variability in food
content of the agent under investigation, its isoforms, the multiple
unknown
interactions between the suspect chemical and the many other ingredients
in the host food, influences of other dietary variables, and even
the pharmacokinetics of the agent in the human body. Nevertheless,
accumulated evidence to date supports a benefit from lycopene in
one form or another, and more so when obtained from whole foods,
in reducing the risk of prostate cancer.73
Selenium
Selenium may have potent anti-cancer properties. Results from laboratory
experiments suggest that selenium-enriched broccoli activates certain
anti-cancer transcription pathways in mouse livers.74 Dog data suggest
that dietary selenium supplementation decreases DNA damage and increases
epithelial cell apoptosis within the aging canine prostate.75
One of several prospective human selenium studies to show a positive
effect on cancer was published in 1996 in JAMA.76 The researchers had
originally chosen only skin cancer but included prostate, lung and
colorectal seven years into the 13-year study because of positive results.
The study included almost 1000 men from regions of the US with mean
plasma selenium concentrations in the lower range of U.S. levels. The
double-blinded, randomized study gave 200 micrograms of selenium as
brewer's yeast, or placebo, daily for 4.5 years and followed
the subjects for an additional 6.4 years. Total cancer incidence (77
cases versus 119), prostate cancers (13 versus 35), colorectal cancers
(8 versus 19), and lung cancers (17 versus 31) were all significantly
lower in the selenium group than in the placebo group. There was no
detectable increase in adverse effects from the supplementation. Because
the study was small and not originally planned for the significant
endpoints and the population was in the lowest range of selenium levels,
it is considered preliminary and in need of verification. Nevertheless,
it strongly suggests a beneficial and safe role for this supplement.
A population study carried out on 212 cases and 233 controls found
a modest negative correlation between serum selenium levels and the
incidence of prostate cancer.77 The inverse association with selenium
was strongest among men with low serum concentrations of a-tocopherol,
another antioxidant.

Essential Fatty Acids
The body synthesizes some fat on its own, but
there are also fats that the body is incapable of manufacturing
known as "essential
fatty acids" or EFAs. They are the omega 3 (alpha-linolenic)
and omega 6 (linoleic) fatty acids. Essential fatty acids are required
constituents of every membrane in the body. Classic signs of deficiency
include depression, mood disorders, memory loss, hyperactivity, anxiety,
dry flaking skin, inflammation, arthritic pain, bursitis, decreased
bone density, easy bruising, muscle spasms, food allergies, fatigue,
increased body fat, sub-clinical and clinical hypoglycemia, hormonal
imbalance and dry hair. EFAs are required to maintain the health
of every living cell in the body. They maintain the fluidity of cellular
membranes, aid in producing and balancing hormones, and play an essential
role in managing inflammation. From essential fatty acids the body
produces many compounds including a group of components known as
prostaglandins. Interestingly, the word prostaglandin comes from
the fact that these products of fatty acid metabolism were originally
found in the prostate gland (prosta - gland - in). Prostaglandins
regulate every organ system in the body. One essential rate-limiting
enzyme in the transformation of EFAs into the important prostaglandins
is d-6-desaturase. Interestingly, zinc deficiency blocks this enzyme.
Other factors that block this enzyme include trans fatty acids, vitamin
B3 and B6 deficiencies, toxic chemicals, alcohol, and some viruses.
A study conducted by medical researchers in Korea established a
connection between BPH, prostate cancer, and essential fatty acids.
Yang et al.78
examined the role of dietary fatty acids in benign and malignant
prostate disease by comparing serum fatty acid levels in normal
controls, patients
with prostate cancer, and patients with BPH. They also looked at
the relative difference in omega 3/omega 6 fatty acid ratios between
the
three groups. The omega 3/omega 6 fatty acid ratios decreased progressively
from control (largest fraction) to BPH to prostate cancer (lowest
fraction) indicating that BPH patients have less omega 3 fatty
acids in their
serum, and prostate cancer patients have significantly less omega
3 fatty acids in their serum. This is a landmark study, as it makes
a
direct correlation between omega 3 fatty acid deficiencies and prostate
disease (BPH and cancer).
Researchers are still not clear on what the entire purpose of the
prostate gland is and the extent of its functions. What the medical
community
does know is that it is a critical gland of the male reproductive
system and that when this gland is diseased in any way, it can cause
a tremendous
amount of pain and suffering for the male patient. After reviewing
the various conditions associated with prostate disease, including
benign prostatic hyperplasia (BPH), prostatitis, and even cancer,
and examining the standard lines of treatment for these conditions,
herbal
and nutrient medicines appear to be an important primary or complementary
therapy for the male patient experiencing sub-optimal prostate health.

Correspondence
Gina L. Nick, PhD, ND
Chief Scientific Officer at Longevity Through Prevention, Inc.
866-587-4622 x702
Fax: 866-587-4622
E-mail: drgina@LTPonline.com
PO Box 6936 Laguna Niguel, California 92607 USA
www.LTPonline.com

References
1. http://www.cdc.gov/nchs/fastats/prostate.htm
2. http://www.cdc.gov/cancer/prostate/prostate.htm
3. McConnell JD et al. Finasteride, an inhibitor of 5 alpha-reductase,
suppresses prostatic dihydrotestosterone in men with benign prostatic
hyperplasia. J Clin Endocrinol Metab.
1992;74(3):505-8.
4. Gormley GJ, et al. The effect of finasteride in men with benign
prostatic hyperplasia. The Finasteride Study Group. N Engl
J Med. 1992; 22;327(17):1185-91.
5. Stoner E. The clinical effects of a 5 alpha-reductase inhibitor,
finasteride, on benign prostatic hyperplasia. The Finasteride Study
Group. J Urol. 1992;147(5):1298-302.
6. Wright J and Lenard L. Maximize your vitality and potency.
Petaluma, CA, Smart Publications, 2000.
7. Cote RJ, et al. The effect of finasteride on the prostate gland
in men with elevated serum prostate-specific antigen levels. Br
J Cancer. 1998;78(3):413-8.
8. Farnsworth WE. Estrogen in the etiopathogenesis of BPH. Prostate 1999;41(4):263-74.
9. NIDDK (National Institute of Diabetes and Digestive and Kidney
Diseases) NIH 1998;98-3012
updated 2000.
10. Williams RM. Environmental clues to prostate cancer. TLfDP 1999;11:144-146.
11. American Medical Association: Health Insight: Prostate
Cancer,
1999.
12. Kao GD, Devine P. Use of complementary health practices by prostate
carcinoma patients undergoing radiation therapy. Cancer.
2000 Feb 1;88(3):615-9.
13. Grieve M. A modern herbal.
Vol 2. New York, NY, Dover Publications, 1971.
14. Tyler VE. Herbs of choice.
Binghamton, NY: Pharmaceutical Products Press, 1994.
15. Lowe FC. Review of recent placebo-controlled trials utilizing
phytotherapeutic agents for treatment of BPH. Prostate 1998;
37(3):187-193.
16. Blumenthal M et al. The complete German commission E monographs:
Therapeutic guide to herbal medicinies.
Trans. S. Klein. Boston MA: American Botanical Council, 1998.
17. Ritchason J. The little herb encyclopedia. Pleasant
Grove, UT, Woodland Health Books, 1995.
18. Ravenna L et al. L et al. Comparison of finasteride, an alpha
5 reductase inhibitor, and various commercial plant extracts, in
vitro
and in vivo. Prostate 1993;22:43-51.
19. Goldmann WH, et al. Saw palmetto berry extract inhibits cell
growth and Cox-2 expression in prostatic cancer cells. Cell
Biol Int. 2001;25(11):1117-24.
20. Gutierrez M et al. Spasmolytic activity of a lipidic extract
from Sabal serrulata fruits: further study of the mechanisms underlying
this activity. Planta Med. 1996
Dec;62(6):507-11.
21. el-Sheik MM et al. The effect of Permixon on androgen receptors.
Acta Obstet Gynecol Scand.
1988;67(5):397-9.
22. Ravenna L et al. Effects of the lipidosterolic extract of Serenoa
repens (Permixon) on human prostatic cell lines. Prostate.
1996; 29(4):219-30.
23. Vacher P et al. Gn-RH agonists in the treatment of prostatic
carcinoma. Biomed Pharmacother.
1995;49(7-8):325-31.
24. Di Silverio F et al. Evidence that Serenoa repens extract displays
an antiestrogenic activity in prostate tissue of benign prostatic
hypertrophy patients. Eur Urol. 1992;21:309-314.
25. Prager N, et al.A randomized, double-blind, placebo-controlled
trial to determine the effectiveness of botanically derived inhibitors
of 5-alpha-reductase in the treatment of androgenetic alopecia. J
Altern Complement Med. 2002 Apr;8(2):143-52.
26. Weisser H and Krieg M. Benign prostatic hyperplasia-the outcome
of age-induced alteration of androgen-estrogen balance. Urology 1997;36(1):3-9.
27. Paubert-Braquet M et al. Effect of the lipid lipidosterolic extract
of Serenoa repens (Permixon) on the ionophore A 23187-stimulated
production of leukotriene B4(LTB4) from human polymorphonuclear neutrophils.
Prostaglandin
Leuko Essen Fatty Acids. 1997:57:299-304.
28. Wilt TJ et al. Saw palmetto extracts for treatment of benign
prostatic hyperplasia: a systematic review. JAMA 1998;280(18):1604-9.
29. Lowe FC. Review of recent placebo-controlled trials utilizing
phytotherapeutic agents for treatment of BPH. Prostate 1998;
37(3):187-193.
30. Carraro JC et al. Comparison of phytotherapy (Permixon) with
finasteride in the treatment of benign prostatic hyperplasia: a randomized
international
study of 1,098 patients. Prostate 1996;29:231-40.
31. Newall CA et al. Herbal medicine:
A guide for healthcare professionals.
London, UK: The Pharmaceutical Press, 1996.
32. Schulz V et al. Rational phytotherapy:
A physician's
guide to herbal medicine. Trans.
Terry C. Telger. 3rd ed. Berlin, Germany: Springer, 1998.
33. Konrad L, et al. Antiproliferative effect on human prostate cancer
cells by a stinging nettle root (Urtica dioica) extract. Planta
Med.
2000 Feb;66(1):44-7.
34. Schottner M et al. Lignans from the roots of Urtica dioica and
their metabolites bind to human sex hormone binding globulin (SHBG).
Planta Med. 1997 Dec;63(6):529-32.
35. Gruenwald J et al. PDR for herbal medicines.
1st ed. Montvale, NJ, Medical Economics Company, Inc., 1998.
36. Schulz V et al. Rational phytotherapy:
A physician's
guide to herbal medicine. Trans.
Terry C. Telger. 3rd ed. Berlin, Germany: Springer, 1998.
37. Wolff H et al. Impact of clinically silent inflammation on male
genital tract organs as reflected by biochemical markers in semen.
J
38. Gansser D et al. Plant constituents interfering with human sex
hormone-binding globulin. Evaluation of a test method and its application
to Urtica dioica root extracts. Z Naturforsch [C].
1995 Jan-Feb;50(1-2):98-104. trans from German.
39. Schmidt K. Effect of radix urticae extract and its several secondary
extracts on blood SHBG in benign prostate hyperplasia. Fortschr
Med.
1983 Apr 21;101(15):713-6. Trans from German.
40. Engelmann U et al. Therapy for benign prostatic hyperplasia with
nettle liquid. Urology 1996;36:287-291.
Trans from French.
41. Dathe G and Shmid H. Phytotherapy for benign prostatic hyperplasia.
Double blind study with Urtica dioica extract. Urology [B]
1987;27:233-236.
42. Vontobel HP et al. Results of a double-blind study on the effectiveness
of ERU (extractum radicis Urticae) capsules in conservative treatment
of benign prostatic hyperplasia. Urologe A. 1985
Jan;24(1):49-51.
43. Metzker M et al. Effects of Sabal-Urtica combination preparations
of benign prostatic hyperplasia (BPH). Urology [B]1996;36:292-300.
44. Sokeland J and Albrecht J. Combination of Sabal and Urtica extract
vs. finasteride in benign prostatic hyperplasia (stages I and II).
Comparison of therapeutic effectiveness in a one year double-blind
study. Urology A 1997;36(4):327-33.
trans from German.
45. German Federal Minister of Justice. German commission E for human
medicine monograph, German Federal Gazette,
no.11.17.01.1991.
46. Foster S et al. Tyler's
honest herbal: a sensible guide to the use of herbs and related remedies.
3rd ed ( 1993) & 4th ed.
(1999). Binghmton NY: Haworth Herbal Press.
47. Chatelain C et al. Comparison of once and twice daily dosage
forms of Pygeum africanum extract in patients with benign prostatic
hyperplasia:
a randomized, double-blind study, with long-term open label extension.
Urology 1999;54(3):472-8.
48. Breza J et al. Efficacy and acceptability of tadenan (Pygeum
africanum extract) in the treatment of benign prostatic hyperplasia
(BPH):a multicentre
trial in central Europe. Curr Med Res Opin 1998;14(3):127-39.
49. Levin RM et al. Beneficial effects of Tadenan therapy after two
weeks of partial obstruction in the rabbit. Neurourol Urodyn 1997;16(6):583-99.
50. Yablonsky F et al. Antiproliferative effect of pygeum africanum
extract on rat prostatic fibroblasts. J Urol 1997;157(6):2381-7.
51. Paubert-Braquet M et al. Effect of pygeum africanum extract on
A23187-stimulated production of lipoxygenase metabolites from human
polymorphonuclear cells. J Lipid Mediat Cell Signal 1994;9(3):285-90.
52. Carani C et al. Urological and sexual evaluation of treatment
of benign prostatic disease using pygeum africanum at high doses.
Arch
Ital Nefrol Androl 1991;63(3):341-5.
trans from Italian.
53. Chatelain C et al. Comparison of once and twice daily dosage
forms of Pygeum africanum extract in patients with benign prostatic
hyperplasia:
a randomized, double-blind study, with long-term open label extension.
Urology 1999;54(3):472-8.
54. Breza J et al. Efficacy and acceptability of tadenan (Pygeum
africanum extract) in the treatment of benign prostatic hyperplasia
(BPH):a multicentre
trial in central Europe. Curr Med Res Opin 1998;14(3):127-39.
55. Levin RM et al. Beneficial effects of Tadenan therapy after two
weeks of partial obstruction in the rabbit. Neurourol Urodyn 1997;16(6):583-99.
56. Yablonsky F et al. Antiproliferative effect of pygeum africanum
extract on rat prostatic fibroblasts. J Urol 1997;157(6):2381-7.
57. Paubert-Braquet M et al. Effect of pygeum africanum extract on
A23187-stimulated production of lipoxygenase metabolites from human
polymorphonuclear cells. J Lipid Mediat Cell Signal 1994;9(3):285-90.
58. Carani C et al. Urological and sexual evaluation of treatment
of benign prostatic disease using pygeum africanum at high doses.
Arch
Ital Nefrol Androl 1991;63(3):341-5.
trans from Italian.
59. http://www.prostate-cancer.org/education/nutrprod/greentea.html
60. Liao S, Hipakka, RA. Selective inhibition of steroid 5? -reductase
[5AR] by tea epicatechin-3-gallate and epigallocatechin-3-gallate.
Biochemical and Biophysical Research Communications 2005;
214(3), 833-838.
61. Feinblatt HM and Gant JC. Palliative treatment of benign prostatic
hypertrophy;value of glycine-alanine-glutamic acid combination. J
Maine M.A. 1958;49:99.
62. Damrau F. Benign prostatic hypertrophy: Amino acid therapy for
symptomatic relief. J Amer Ger Soc 1962;10(5):426-30.
63. Cuervo B et al. Clinical study of a phytosterol extract of prunus
arborea and 3 amino acids: glycine, alanine, and glutamic acid. Arch
Esp Urol 1978;31(1):97-8. trans from
Spanish.
64. Maekawa M et al. Clinical evaluation of Cernilton on benign prostatic
hypertrophy – a multiple center double- blind study with paraprost.
Hinyo Kiyo 1990 ;36(4):495-516.
65. Zaichick VY et al. Zinc in the human prostate gland: normal hyperplastic
and cancerous. Int Urol Nephrol 1997;29(5):565-74.
66. Costello LC and Franklin RB. Novel role of zinc in the regulation
of prostate citrate metabolism and its implications in prostate cancer.
Prostate 1998:35(4):285-96.
67. Lagiou P et al. Diet and benign prostatic hyperplasia: a study
in Greece. Urology 1999;54(2):284-90.
68. Costello LC and Franklin RB. Novel role of zinc in the regulation
of prostate citrate metabolism and its implications in prostate cancer.
Prostate 1998:35(4):285-96.
69. Brys M et al. Zinc and cadmium analysis in human prostate neoplasms.
Biol Trace Elem Res 1997;59(1-3):145-52.
70. Zaichick VY et al. Zinc concentration in human prostatic fluid:
normal, chronic prostatitis, adenoma and cancer. Int Urol
Nephrol 1996;28(5):687-94.
71. Habib FK et al. Androgen concentrations in expressed prostatic
secretions. Urol Res 1992;20(4):281-4.
72. Leitzmann MF, et al. Zinc supplement use and risk of prostate
cancer. J Natl Cancer Inst.
2003;95(13):1004-7.
73. Hadley, CW, et al. Tomatoes, Lycopene, and Prostate Cancer: Progress
and Promise. http://www.lycored.com/pdf/clinton.pdf (July
2005: Link dead. Article also found at http://www.ebmonline.org/cgi/content/full/227/10/869)
74. Zeng, H, CD Davis, and JW Finley. Effect of selenium-enriched
broccoli diet on differential gene expression in Min mouse liver.
Experimental
Biology 2003 meeting. April 11-15. San Diego. http://www.sciencenews.org/articles/20030503/food.asp
75. Waters DJ et al. Effects of dietary selenium supplementation
on DNA damage and apoptosis in canine prostate. Journal of
the National Cancer Institute 2003;95:237-241.
76. Clark, LC et al. Effects of selenium supplementation for cancer
prevention in patients with carcinoma of the skin. A randomized controlled
trial. Nutritional Prevention of Cancer Study Group. JAMA.
1996; 25;276(24):1957-63.
77. Vogt TM et al. Serum selenium and risk of prostate cancer in
U.S. blacks and whites. International Journal of Cancer 2003;
103:664-670.
78. Yang YJ et al. Comparison of fatty acid profiles in the serum
of patients with prostate cancer and benign prostatic hyperplasia.
Clin
Biochem 1999; 32(6):405-9.
|





