Page 1, 2, 3
This is why, in addition to the loss of tumor suppressive functions, mutant P53 increases oncogenic function, contributing to carcinogenesis, tumor progression, invasion, metastases, and drug resistance.37 Reversing P53 mutation to normal, wild-type function is associated with increasing apoptosis and decreasing tumor progression.38 I have demonstrated over the past years how some selected dietary agents and bioactive compounds can reverse mutant P53 to a normal WT-P53 tumor suppressor gene in numerous cancers and increase the destruction of cancer cells.39,40 (For more references read: Oncogenic activation in prostate cancer progression and metastases: Molecular Insight and Future Challenge. Journal of Carcinogenesis. (April 17, 2012) available online.)
Anti-Apoptotic Players in Prostate Cancer
BCL-2 plays a key role in earlier cancer development. BCL-2 is the founding member of the BCL-2 family of regulator proteins that regulate cell death (apoptosis) via the mitochondria by either inducing apoptosis (BAX gene) or inhibiting apoptosis (BCL-2).41 BCL-2 is considered an important anti-apoptotic gene and protein that blocks programmed cell death.42 BAX is the homologue of BCL-2, a pro-apoptotic protein that resides in the cytosol. Under stimuli, BAX is transcriptionally activated by P53, leading to its subsequent insertion into the outside membrane forming mitochondrial pores43 and increasing its permeability,44 which leads to the release of apoptotic factors such as cytochrome C that activate the apoptotic cascade.45,46
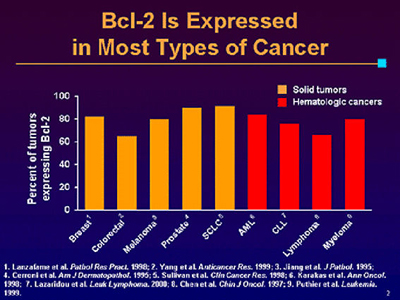
BCL-2 is located in the inner membrane of mitochondria and, when overexpressed, blocks the apoptotic protein BAX, preventing the formation of mitochondrial pores inhibiting the release of the enzyme cytochrome C, thus either inhibiting or reducing the death of cells.47 Both in-vitro and in-vivo studies have established that BCL-2 and other anti-apoptotic members of its family are significantly upregulated for the progression of prostate cancer and in aggressive prostate cancer phenotypes.48 Overexpressed BCL-2 in human prostate cancer confers resistance and prolongs cell survival, increasing progression of metastases despite chemotherapy or exposure to radiation therapy.49
 According to an article published in Lancet magazine about prostate cancer and radiotherapy (June 2007;8:475-478), conventional radical external-beam radiotherapy is limited to a dose of 64-70 Gy in 1.8-2.0 Gy fractions because of the risk of long-term toxic effects to the bladder and rectum. However, within five years of being treated with this type of radiotherapy, up to 33% of patients will relapse, which seems unacceptable. According to an article published in Lancet magazine about prostate cancer and radiotherapy (June 2007;8:475-478), conventional radical external-beam radiotherapy is limited to a dose of 64-70 Gy in 1.8-2.0 Gy fractions because of the risk of long-term toxic effects to the bladder and rectum. However, within five years of being treated with this type of radiotherapy, up to 33% of patients will relapse, which seems unacceptable.
Some studies that include a large patient population demonstrated that BCL-2 expression levels were of major significance in predicting clinical outcome after brachytherapy of prostate cancer patients. BCL-2 was significantly higher in prostatic tumors resistant to radiotherapy compared with radiation responders. Recent studies have linked the expression of BCL-2 to the development of the hormone resistant phenotypes of advanced prostate cancer.49 BCL-2 is negatively regulated by P53 to trigger apoptosis.50 The response to cancer cells to exposure of radiotherapy is not only associated with BCL-2 expression but P53 tumor suppressor.51 Therefore, we understand that loss of P53 function may negatively influence the expression of BCL-2 in prostate cancer.52
The BCL-2/BAX ratio is considered a predictive marker for the response to chemotherapy or radiation therapy in prostate cancer.53 Where other oncogenes are activated, BAX is lost with a poor ratio. BCL-2 overexpression may confer an additional growth advantage to tumors.
Prostate cancer can become resistant to apoptosis during the progression of the disease and metastasis via multiple molecular mechanisms. BCL-2 is not highly expressed in early local prostate cancer but is highly expressed in metastatic tumors. It can be triggered in several ways such as inflammation and by NFKB, which, in fact, transcriptionally regulates BCL-2.54
As discussed previously, C-Myc is frequently amplified in prostate cancer; and there is cross talk or intervention between C-Myc and BCL-255 that increases expression. Often, this is the result of excessive chemotherapy.
Overall, BCL-2 overexpression is associated with progression of prostate cancer, tumor resistance to chemotherapy/radiation, and a poor prognosis. As a result, it may prove to be an important independent or adjuvant predictor of resistance to apoptosis during conventional treatment and needs to be targeted.
The Role of Survivin in Prostate Cancer
Survivin is a new anti-apoptotic protein with bifunctional function implicated in the protection of apoptosis and modulation of cell division,56 playing a crucial role in cancer development and metastatic potential. Survivin is a strong inhibitor of apoptosis protein,57 strongly expressed in embryonic and fetal organs but undetectable in differentiated normal tissue. It is upregulated in 84% of all cancers including breast, colon, bladder, pancreas, lymphoma, and prostate.58
Therefore, survivin becomes a new cancer biomarker and an attractive molecule for early detection and prognosis of cancers such as breast, prostate, bladder, and colorectal cancer.59,60
The primary function of survivin is the inhibition of apoptosis under stimuli, but it also regulates mitosis, which is associated with cell cycle and carcinogenesis.61,62 Survivin in the mitochondrial membrane is rapidly released in the cytosol in response to apoptosis and inhibits apoptosis either directly or indirectly by interfering with the function of caspase 3, caspase 7, and caspase 9.63 A family of 14 cysteine proteases required for the initiation and execution of apoptosis may also be a caspase-independent way by interaction and cooperation with Hepatitis B interacting protein (HB x IP) or XIAP to inhibit apoptosis.64 Survivin expression enhances the metastatic potential of the tumor, using HBxIP and XIAP and even NF-kappa B as co-factors, and increases the resistance of cancer cells.
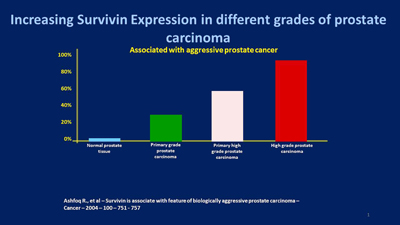
The expression of survivin increases gradually from normal prostate tissue, to primary low-grade prostate carcinoma, with the highest expression observed in the foci of prostate cancer metastasized to the lymph nodes.65,66 This pattern of gene expression suggests that survivin is associated with disease progression and accelerated rate of recurrence67 and increased resistance to therapy.68,69 Therefore, it makes survivin an attractive target for prostate cancer therapeutics or prevention of recurrence, as I am going to show with examples of clinical cases.
Survivin gene expression is transcriptionally regulated by wild type P53.70 The loss of P53 function not only deregulates survivin gene expression but also BCL-2 gene expression with a stronger anti-apoptotic mechanism. Although they differ,71 both confer a more resistant type of cancer cells and a bad prognostic.72
Hypoxia and Survivin Expression
Hypoxia increases survivin expression and promotes angiogenesis, proliferation, and metastatic invasion. Hypoxia is a potential prognostic and predictive factor in prostate cancer (and other cancers) that significantly correlates with the Gleason Score73 and leads to the selection of aggressive cancer, decreased sensitivity to apoptosis, and an increased clinical stage.74 Hypoxia may have a negative effect by increasing angiogenic potential and metastatic capability leading to tumor expansion and metastatic progression, such as in advanced stage prostate cancer.75
The presence of hypoxic regions in a solid tumor is a common phenomenon and results in dramatic alteration in gene expression76 that increases the level approximately threefold,77 which in turn increases the resistance and aggressiveness of prostate cancer cells. Upregulated survivin78-80 was shown to favor angiogenesis, increasing vesicular endothelium growth factor (VEGF) and fibroblast growth factor (FGF), also identified in prostate cancer.81-83
Survivin is also strongly upregulated in angiogenically stimulated endothelium in-vitro and in-vivo and protects endothelial cells from apoptosis, not only during the proliferative phase of angiogenesis84 but also during the remodeling phase mediated by angioprotein.
Vesicular endothelium growth factor (VEGF) is a potent stimulator of angiogenesis that tumors need to expand. Cancer cells can express VEGF to metastasize, which also correlates with P53 mutation in hypoxic tissue.85
Highly expressed survivin in prostate cancer induces resistance to treatment86 and is associated with aggressive prostate carcinoma.87 A new avenue is emerging to increase the effectiveness of anticancer agents by targeting survivin at the clinical level.88,89 On the other hand, survivin expression usually is correlated with the BCL-2 expression and often the loss of P53 that strongly increases the resistance of cancer cells. It may serve as a good predictive factor to determine the response to chemotherapy and radiotherapy with cancer patients. It may also reduce the risk of metastases diffusion by targeting P53, BCL-2, and survivin.90
Evidence has shown that restoring P53 function not only results in increasing apoptosis in prostate cancer91 but downregulates BCL-2 and survivin gene expression, increasing the effectiveness of chemo/radiation therapy. Targeting apoptosis in prostate cancer is now emerging as a novel anticancer therapy.92
In summary, key points regarding survivin over-expression are as follows:
- Absent in normal tissue, highly expressed in cancer tissue;
- Can be used as a biomarker;
- Released by mitochondria under pro-apoptotic stimuli;
- Inhibits caspases 3-7-9;
- Enhances angiogenesis;
- Is associated with chemotherapy resistance and increased rate of tumor recurrence;
- Indicates poor outcome; and
- Predicts prostate cancer recurrence.
The Protocol for Prostate Cancer
New avenues in the apoptosis pathway, modulation of inflammatory mediators, oncogene expression, and the immune system offer excellent opportunities for approaching the disease of cancer differently. These new approaches can render antineoplasic agents more efficient, avoid unnecessary treatments and their side effects, and prevent the progression of the tumor to advanced metastases disease.
It makes an overwhelming difference when you do blood testing of your patient because you realize that patients with the same cancer have different results concerning the activity of tumor suppressors and oncogenes and different ratios between the pro-apoptotic and anti-apoptotic proteins, which permit you to make your prognostic. Another test we use is the Tumor Marker-2 – Pyruvate Kinase that indicates the metabolic activity of the tumor and supports the antigen PSA test. With these tests, you can follow the recovery of patients. Each patient needs a personalized or individualized treatment based on the results of the blood tests, not to mention the age, the general status, and family background.
My protocol for the treatment of prostate cancer has been developed and refined over many years of practice and observation. As a cornerstone, we use plant-derived dietary agents and other ingredients that are immunomodulatory with multi-targeting properties against cancer. These agents are safe, increasing chemotherapy/radiation effectiveness while reducing unpleasant side effects. Also, this approach has proven efficiency in treatment without conventional therapy.
Page 1, 2, 3 |
![]()
![]()
![]()
![]()







