|
Page 1, 2, 3
This article was reviewed by Dr. Joseph Brenner, medical oncologist, head of the Oncology Institute, Wolfson Hospital, and director of the New-Hope Medical Center for Integrative Cancer Treatment in Holon, Israel. Dr. Brenner was trained in chemotherapy and radiotherapy at the Memorial Sloan Kettering Medical Center in New York City.
Abstract
Cancer, namely prostate cancer is a disease of cell cycle regulation and checkpoint causing uncontrolled cellular proliferation, tumor promotion, apoptotic failure, angiogenesis, tumor growth and metastases invasion.
The P53 tumor suppressor gene is the central hub in the cellular growth pathway; but other genes transcriptionally controlled by P53 and other transcription factors are mostly involved in prostate cancer including Myc, Pten, TGFB, BcL2, and especially survivin, an inhibitor of apoptosis. Survivin is highly expressed in prostate cancer with a metastasis condition and is associated with poor patient prognosis and resistance to chemotherapy. Up-regulated BcL2, survivin, and failure or P53 mutation are also associated with metastasis invasion and disease progression.
 Restoring P53 function and targeting apoptotic and anti-apoptotic players together with the activation of immune cells, as natural killer cells (NK cells), is now emerging as a novel anticancer therapy. The clinical application of blood testing for apoptotic and anti-apoptotic genes remains an important goal from the perspective of establishing a personal therapy for each patient. Restoring P53 function and targeting apoptotic and anti-apoptotic players together with the activation of immune cells, as natural killer cells (NK cells), is now emerging as a novel anticancer therapy. The clinical application of blood testing for apoptotic and anti-apoptotic genes remains an important goal from the perspective of establishing a personal therapy for each patient.
Many lines of study have suggested that some natural products may have the ability to target a cell's signaling pathway, downregulate BcL2 and survivin, upregulate the Bax gene, and restore P53 function. Tegaran (Zhen Hua), a compound made from unmodified fermented soya extract that contains isoflavones, protease inhibitors, saponins, phytic acid, and genistein, has shown therapeutic capacity for adjuvant treatment of solid tumors. Zhen Hua is a most effective agent that targets transcription factors, decreases NF-KB activity, VEGF, and MDR1. It also targets BcL2, Bax, and MDR1 expression in various cancer cell lines.
Curcumin, the second compound, is a yellow coloring ingredient derived from Curcuma longa L.; it is one of the most extensively investigated and well-defined chemopreventive agents. A large variety of pharmacological properties, including anti-inflammatory, antioxidant, and anti-cancer, has been demonstrated in preclinical animal and pilot human studies. Curcumin arrests the cell cycle at GO/G1 or G2 (same as most chemotherapy) inducing apoptosis. Curcumin is known to target most of the hallmarks of cancer, downregulate BcL2, survivin, VEGF, Cox2, and NF-kB and increase immune cell activity.
The third compound is a liquid cartilage extract (LCE) having properties that target most of the angiogenesis mechanisms including VEGF and MMPs. Several studies reveal that LCE interferes with VEGF binding and signaling while inhibiting MM 2, 9, and 12, which are often overexpressed in tumors and involved in both migration of endothelial cells and tumor invasion. Other secondary but interesting dietary compounds like green or black propolis and pomegranate also have shown some interesting ability as a support in the protocol of prostate cancer.
Introduction
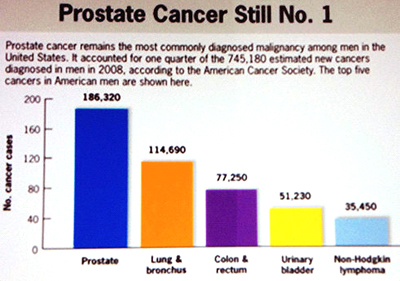
In North America, prostate cancer is the most commonly diagnosed cancer in men; before, it used to be lung cancer. In 2012, 177,489 men were diagnosed with prostate cancer in United States and 27,244 men died from prostate cancer (US cancer statistics working group 1999-2012 – CDC and NCI, 2015). In 2012, 1.1 million men were diagnosed with prostate cancer worldwide. A larger increase of prostate cancer is expected by 2020 (CDC), not only in United States but in most industrial countries. Prostate cancer is the cause of death in 15% of patients diagnosed with the disease in the USA but in 50% of those diagnosed in England. The difference is most probably due to huge screening efforts in the USA by PSA detection in healthy individuals and early detection of the disease due to screening (2011 Meeting of United Kingdom Association of Cancer Registries and National Cancer Intelligence Network (NCIN). Abstract 86. Presented June 16, 2011).
Most prostate cancers are asymptomatic and slow growing; however, a third of the cases are more aggressive and fast developing.1 Prostate metastasis spreads relatively quickly to other parts of the body, particularly lymph nodes, rectum, bones, and brain, ultimately leading to death. Treatment includes a combination of surgery, hormonotherapy, radiation therapy, and chemotherapy that may be effective for the cure of localized disease; but there is no effective treatment strategy for metastasis, thus prostate cancer remains painful and often incurable.2 Aggressive prostate cancer accounts for more cancer-related mortality than any other cancer including lung cancer. Despite advances in local treatment, approximately 25% of radically treated patients will develop disease progression3; and in 10-20% of present cases, starting at the primary diagnosis of bone metastases, the use of hormones and or chemotherapeutic drugs could only extend the lives of these men or those with other advanced cancer by months or a few years at best. Prostate cancer tumor progression and aggression can be secondary to genetic and epigenetic changes4 through the activation of oncogenes and inactivation of tumor suppressor genes, to an angiogenic switch5 and immune evasion.6
Recent advances into the molecular function of pro-apoptotic genes and inhibitors of apoptosis open a new door to potential therapeutic targeting opportunities for the treatment of prostate cancer, while prevention and diagnostics at an early stage remains a main goal in naturopathic oncology.7
Prostate Cancer
Evidence indicates that chronic inflammation promoted by oxidative stress and dietary factors8 damages the DNA genome along with mitochondrial DNA (mtDNA). Several reports have highlighted the link between metabolic alteration and mtDNA mutation in prostate cancer and associated these factors to the development and progression of the disease.9 The progressive loss of cell cycle regulation, causing uncontrolled cellular proliferation, is today attracting more attention to the point where we say that cancer is a disease of the cell cycle.10
According to an article published in Lancet (August 2004), "Gene Therapy for Prostate Cancer," selective killing of prostate cancer can be achieved by the manipulation of cell-cycle control and apoptosis through tumor suppressor genes, such as P53 and BAX. (See my conference presentation, "How to understand and treat cancer from a molecular basis" at http://www.slideshare.net/SheldonStein/how-to-understand-treat-cancer-w-molecular-markers.)
Evasion from apoptosis is critical for tumor growth and a main hallmark in cancer.11 Abnormal cells accumulate mutations, become resistant, and start to proliferate. Metastatic cancer cells can escape from the primary tumor through the loss of cell-adhesion, induction of cell mobility, and start to invade the surrounding tissue. They enter into blood or lymphatic circulation, travel to distant organs and establish a new tumor that can keep dormant over several years12 or reactivate after a few months inducing angiogenesis to grow again.
In fact, there are several primary contributors in the emergence of androgen-independent metastatic prostate cancer such as activation of survival pathways, including apoptosis, suppression of anoikis resistance,13 and increased neovascularization. Of course, evasion from immune surveillance is also a contributor.
Therefore, as I will show in this article, targeting apoptotic players and angiogenesis is of vital therapeutic significance since resistance to apoptosis plays a key role in therapeutic failure to standard treatment strategies and plays a paramount role in tumor progression and disease recurrence.14,15
Several basic elements are involved in the malignant process and are considered hallmarks of cancer:
- Evading apoptosis (P53 tumor suppressor),
- Overexpressed BCL2 gene,
- Activated P13K/AKT pathway,
- Loss of E. Cadherin,
- Overexpressed Survivin gene, and
- Sustained angiogenesis.
Without these hallmarks, there would not be any cancer, a very complex disease. We have a new promising avenue that leads to new diagnostics and more effective treatments for cancer.
Molecular Basis of Prostate Cancer
Although there is a myriad of genes implicated in cancer, we are going to discuss briefly some of the most important ones associated with prostate cancer. Somatic genetic and epigenetic alterations occur in prostate cancer such as the loss of the tumor suppressor P53 network, PTEN, C-Myc, apoptosis, invasion, angiogenesis and metastases. P53 tumor suppressor is situated at the crossroad of a network of signaling pathway16 composed of subway stations where each gene is connected with each other and engage in cross talk. P53 is a central hub in the cellular growth pathway and cooperates with other genes and transcription factors, including Myc, PTEN, NF-kappaB, FG, and TGFB .
C-Myc is one of the most commonly studied oncogenes, being a regulator gene that is coded for transcription factors associated with growth control, cell cycle, differentiation and apoptosis.17 C-Myc is frequently amplified in prostate cancer patients. FISH analysis identified overexpression of C-Myc in 9% of primary tumors but 75% in advanced cancer patients.18,19 There is cross talk between C-Myc and P53; C-Myc increases P53 activity in the case of apoptosis, and P53 mediates C-Myc activity but not in the case of mutant P53.20,21
PTEN is another tumor suppressor frequently inactive in prostate cancer,22,23 one of the most common genetic alterations in prostate cancer. Loss of PTEN is needed for cancer progression to occur in prostate cancer24 and a common event in treatment. Resistant and poorly differentiated prostate cancer leads to constitutive activation of the P13/AKT, a regulator of the cell cycle and directly related to cellular proliferation and cancer, including prostate cancer.25 Both PTEN inactivation and P13/AKT hyperactivity are hallmarks of aggressive prostate cancer, resistant to chemotherapy.26 There is cross talk between PTEN and P53 tumor suppressor; P53 can regulate PTEN at the transcription level, and PTEN can regulate P53 protein level activity27 or can be regularized by MDM2 (a negative regulator of tumor suppressor P53). Therefore, PTEN also sensitizes cells to chemotherapy28 by increasing apoptosis.
P53 mutation downregulates C-Myc and other transcription genes associated with apoptosis, angiogenesis and immune system such as the transforming growth factor beta (TGFB),29 an essential regulator of cellular physiologic processes including proliferation, differentiation, cell survival, apoptosis, angiogenesis, and immune response. TGFB plays a positive role as a tumor suppressor by promoting apoptosis at early stages of the cancer process, facilitated by the presence of WT P53 but not with the presence of mutant P53 that inhibits the TGFB tumor suppressor. Upregulation of TGFB can stimulate invasion, angiogenesis, and metastases.30 Elevated TGFB levels in a patient's serum provide a significant prognostic value for highly aggressive metastatic diseases and poor patient prognosis in prostate cancer.31 For this reason, direct targeting of TGFB receptors also provides a promising therapeutic avenue in the inhibition of tumor progression in clinical applications.32
Another example is the transcriptional cross talk between the P53 tumor suppressor and the nuclear factor kappa B (NFKB), a major mediator of inflammation and suspected to be involved in cancer development, progression, and activation in prostate cancer.33 NFKB is associated with angiogenesis, activating VEGF, the protease enzymes MMP-9, apoptosis, and immunosuppression. Considering the deregulation of NFKB and P53 pathways in numerous cancers, it is not surprising that an extensive cross talk between both pathways exists at various levels.34 Normal P53 can suppress the activity of NFKB35; but mutant P53, to the contrary, enhances NFKB, constitutively activated in human prostate adenocarcinoma. This correlates with resistance to various chemotherapeutic agents, such as paclitaxel, in prostate cancer and disease progression. Targeting NF-kappa B in association with chemotherapy strongly enhances the apoptotic potential of the chemotherapy and is associated with suppression of angiogenesis and metastases in human prostate cancer.36
Page 1, 2, 3
|
![]()
![]()
![]()
![]()






