|
Added online November 2016
Introduction
Cancer is a fatal and complex disease stemming from genetic aberrations and epigenetic changes, resulting in atypical, uncontrolled cell overgrowth. The World Health Organization (WHO) estimates that 7.5 million patients worldwide die from this disease every year.1 Although several conventional therapeutic options, such as surgery, external radiotherapy, chemotherapy, and immunotherapy, are currently available and have initial high response rates, most deaths are attributed to a subsequent relapse or recurrence of the disease. In fact, even though chemotherapy is currently one of the most common therapeutic options, the outcomes have been rather disappointing or unsatisfactory, with severe side effects.2 Thus, establishing more effective and safer therapeutic modalities for these cancer patients is urgently demanded.
To improve the efficacy of chemotherapy, we have been exploring combination therapies using chemotherapeutic drugs and natural agents extracted from herbs, plants, mushrooms, seeds, fruits, and so on. Among them, we were particularly interested in "D-fraction" (DF), a bioactive extract from maitake mushroom (Grifola frondosa). This DF is the protein-bound polysaccharide or proteoglucan, consisting of a bioactive component known as b-glucan.3 DF has been commercially available for a variety of medical and scientific studies, and numerous studies have shown its immunomodulatory and antitumor activities in an animal model.4,5 In addition, the antiviral effect of DF was demonstrated on hepatitis B and also confirmed on human immunodeficiency virus (HIV)/AIDS in anti-HIV drug screening tests conducted by the US National Cancer Institute in 1992.6,7 Thus, DF appears to be an interesting and promising natural agent that deserves further study, particularly on its antitumor activity.
Moreover, regarding the safety issue of DF, the US Food and Drug Administration (FDA) has exempted DF from a phase I study of toxicology tests. The FDA has also approved DF for the Investigational New Drug (IND) application for a phase II pilot study on patients with advanced breast and prostate cancer.8 A randomized clinical trial of DF on healthy subjects (noncancerous participants) has further confirmed no palpable ailments or adverse effects on any participants.9 Therefore, all these facts would support the safety of DF in cancer patients as well as healthy people.
Accordingly, we investigated if the efficacy or anticancer effects of various chemotherapeutic drugs on four types of human cancer cells might be potentiated in combination with DF. Those cells included breast, lung, stomach, and colon cancers. Interestingly, as it has been proposed that vitamin C (VC) could modulate bioactivity of DF, we also examined if such a combination might have antiproliferative effect on a total of 10 cancer cells, additionally including 3 urologic and 3 other breast cancer cell lines that were known to be drug resistant.10
Results
Chemotherapeutic Drugs and DF: Effects on Growth of Various Cancer Cells
A total of 10 human cancer cell lines were used in this study: the 4 types of breast cancer cell lines, MDA-MB231, MCF-7, ZR-75-1, and T-47D; the lung cancer A549 cells; the stomach cancer AGS cells; the colon cancer Caco-2 cells; and 3 urologic cancer cell lines, prostate cancer PC-3, bladder cancer T24, and renal cell carcinoma ACHN. The effects of chemotherapeutic drugs were first examined on 4 cancer cell types, including MDA-MB231 (breast), A549 (lung), AGS (stomach), and Caco-2 (colon). Specified drugs tested were clinically being used in each cancer case: paclitaxel (PTX) for breast cancer, cisplatin (CPL) for lung cancer, and 5-fluorouracil (5-FU) for stomach and colon cancers. For experiments, MDA-MB231, A549, AGS, and Caco-2 cells were cultured with PTX (0–30 nM), CPL (0–100 µM), 5–FU (0–300 ng/ml), and 5-FU (0–300 ng/ml), respectively. The same experiments were also carried out with combinations of PTX, CPL, or 5-FU and DF (300 µg/ml). Cell growth was then assessed at 72 hours. It should be noted that DF (300 µg/ml) by itself had little effect on all 4 cancer cells in our pilot study (data not shown). Additionally, the concentrations of these drugs used were the estimated maximum physiologically tolerable levels.
MDA-MB231 Study (Figure 1A):
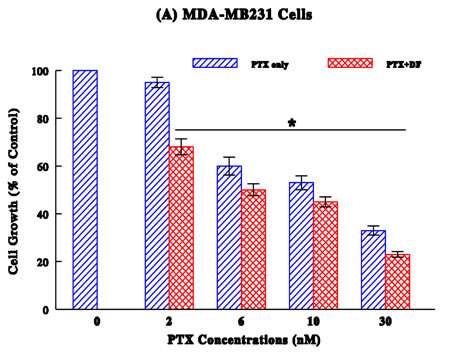
PTX by itself has the significant antiproliferative or growth-inhibitory effect (p < 0.05), leading to a ~40, ~50%, and ~70% growth reduction at 6, 10, and 30 nM, respectively. However, its combination with DF (300 µg/ml) slightly improved such antiproliferative activity (i.e., only ≤10% increase in the growth reduction).
A549 Study (Figure 1B):
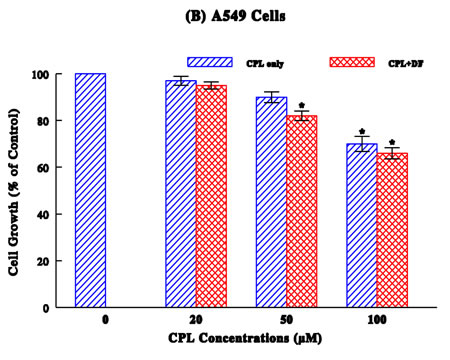
No significant growth inhibitory effects were observed with CPL concentrations up to 100 µM with a ~30% growth reduction. Even its combination with DF (300 µg/ml) barely showed any improved growth reduction (up to only 34%).
AGS Study (Figure 1C):

5-FU didn’t seem to be very effective and its maximum concentration of 300 ng/ml tested led to a ~20% growth reduction. However, its combination with DF (300 µg/ml) was capable of significantly enhancing antiproliferative activity, resulting in a ~24% and ~42% growth reduction from a ~10% and ~20% reduction with 5-FU (200 and 300 ng/ml) alone, respectively.
Caco-2 Study (Figure 1D):
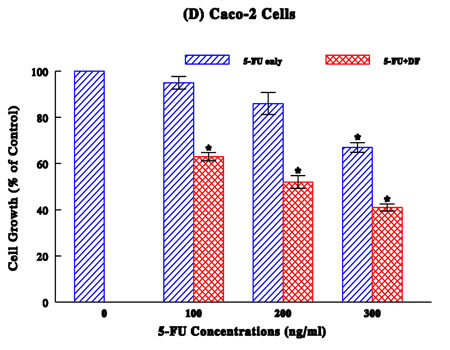
No significant growth reduction with 5-FU was seen up to 300 ng/ml with a ~33% growth reduction. Yet, its combination with DF (300 µg/ml) led to the enhanced growth reduction: even 5-FU (100 ng/ml) with no apparent growth-inhibitory effect has induced a ~37% growth reduction with DF, while combinations of 200 and 300 ng/ml 5-FU (with a ~14% and ~33% growth reduction, respectively) and DF resulted in a ~50% and ~60% growth reduction, respectively.
Taken together, these results suggest that DF appears to enhance antiproliferative activity of chemotherapeutic drugs to some extent on various cancer cell types.
Synergistic Antiproliferative Effects of Combination of DF and VC
As VC has been implied to enhance bioactivity of DF, we examined whether a combination of DF and VC could exhibit any antiproliferative activity.8 In addition to 4 cancer cell types above, 3 common urologic cancer cell lines, prostate cancer PC-3, bladder cancer T24, and renal cell carcinoma ACHN; and 3 additional breast cancer cell lines, MCF-7, ZR-75-1, and T-47D, were also included in this study. Since these 6 cancer cells exhibited a drug-resistant nature in our separate studies, it was of interest to examine whether such DF/VC combination might have antiproliferative effect on them. Accordingly, all 10 different cancer cells were cultured with combination of DF (300 µg/ml) and VC (200 µM) and cell growth was assessed at 72 hours. It should be noted again that neither DF (300 µg/ml) nor VC (200 µM) alone had any effects on all these cancer cells (data not shown). The results are summarized in Table 1.pdf
Overall, the combination of DF and VC was highly potent, inducing a ≥90% growth reduction in nearly all cancer cells except for a ~70% growth reduction in lung cancer A549 cells. This also indicates that DF and VC may work synergistically to induce such a severe growth reduction. Therefore, DF/VC combination appears to be more potent to induce a profound growth reduction in all 10 cancer cells than any combinations of chemotherapeutic drugs and DF (Figures 1A–D).
Comment
In an attempt to improve the poor efficacy of chemotherapy on various human cancers, we investigated if a variety of chemotherapeutic drugs could be potentiated with bioactive D-fraction (DF) extracted from maitake mushroom. As DF with active component of proteoglucan (or beta-glucan) has been shown to have antitumor activity with few side effects, it was used in our combination study (with various drugs).3-5
Several common human cancers, such as breast, lung, stomach, colon, and urologic (prostate, bladder, and kidney), were employed and a dose-dependent study showed the drug concentration-dependent growth reduction in a cancer-specific manner (Figures 1A–D). As all these growth reductions (relative to respective controls) were confirmed to be statistically significant (p < 0.05), drugs tested here have the growth-inhibitory or antiproliferative effects to certain extent.
To improve the drug efficacy, we examined if DF would enhance or potentiate the efficacy of those drugs. Such study showed that all drugs tested were somewhat potentiated with DF, resulting in the marginal (4%) to significant (26%) higher growth reductions (than those with drugs alone). It appears that DF could help improve the efficacy of all drugs, although it may work better with certain drugs than others.
Additionally, as an alternative and unique approach, we also examined if a combination of DF (300 µg/ml) and VC (200 µM) may also have some antiproliferative effect on those cancer cells. Remarkably, such a DF/VC combination was capable of inducing a drastic ≥90% growth reduction in 9 cancer cell lines except for a ~70% growth reduction in 1 cancer cell line (Table 1). These results are thus compelling and demonstrate that the DF/VC combination is superior to any drug alone or any combinations of drugs and DF tested, implying their possible clinical application.
Although the potency of DF/VC combination is the most interesting finding in this study, it is yet preliminary, and especially how DF/VC combination would induce a severe growth reduction in various cancer cells needs to be elucidated. To unveil such an antiproliferative mechanism, further studies are currently under way in our laboratory.
In conclusion, the present study suggests that D-fraction appears to have a great potential as an adjuvant agent that can be combined with certain chemotherapeutic drugs or with vitamin C for the more effective treatment of various human malignancies.
Acknowledgement
We would like to thank Mr. Mike Shirota (Mushroom Wisdom Inc.) for a generous gift of D-fraction and his devoted support.
Sensuke Konno, PhD
Department of Urology
New York Medical College
Munger Pavilion 4th Floor
Valhalla, New York 10595
sensuke_konno@nymc.edu

Notes
1. Maiti AK. Genetic determinants of oxidative stress-mediated sensitization of drug-resistant cancer cells. Int J Cancer. Epub 2011 July 25; doi:10.1002/ijc.26306.
2. Fung-Kee-Fung M, Oliver T, Elit L, Oza A, Hirte HW, Bryson P. Optimal chemotherapy treatment for women with recurrent ovarian cancer. Curr Oncol. 2007;14:195–208.
3. Mizuno T, Zhuang C. Maitake, Grifola frondosa: pharmacological effects. Food Rev Int. 1995;11:135–149.
4. Adachi K, Nanba H, Kuroda H. Potentiation of host-mediated antitumor activity in mice by b-glucan obtained from Grifola frondosa (maitake). Chem Pharm Bull (Tokyo). 1987;35:262–270.
5. Hishida I, Nanba H, Kuroda H. Antitumor activity exhibited by orally administered extract from fruit body of Grifola frondosa (maitake). Chem Pharm Bull (Tokyo). 1988;36:1819–1827.
6. Wu S, Zou D, Han SH et al. Therapeutic effect of Grifola polysaccharides in chronic hepatitis B. In: International Symposium on Production and Products of Lentinus Mushroom, Programme and Abstracts. Qingyuan, China; November 1–3, 1994.
7. Developmental Therapeutics Program, National Cancer Institute: In-vitro anti-HIV drug screening results. Washington, DC: National Science Council F195001; January 1992.
8. Maitake Products Inc. D-fraction obtained IND for clinical study. Corporate publication. Paramus, NJ; February 1998.
9. Glauco S, Jano F, Paolo G, Konno S. Safety of maitake D-fraction in healthy subjects: assessment of common hematologic parameters. Altern Complement Ther. 2004;10:228–230.
10. Morishige F. The role of vitamin C in tumor therapy (human). In: Meyskens FI Jr, Parasad KN, eds. Vitamins and Cancer: Human Cancer Prevention by Vitamins and Micronutrients. Clifton, NJ: Humana Press; 1986:399–427.


|
|



![]()
![]()
![]()





