|
Abstract
Breast cancer is the most common malignancy in the Americas, especially among Northwestern European women. It remains the number 1 cancer killer. Despite progress in oncology, benefits of treatment, the gold standard of palliative therapy, radiation, and surgery, survival rates for patients with metastatic breast cancer continue to remain poor. Recurrence is still too high. 70% of women with primary cancer are subject to disease recurrence within the first 2 years, no matter which surgery or treatment they receive.1 Disease survival rates and recurrence depend upon the disease stage, but a new avenue for diagnosing cancer, focusing on the P53 apoptotic pathway and P53 mutation, seems to correlate with breast cancer recurrence.
Our Experience with P53 and Other Targeting Molecular Markers.
A few years ago, we started working with P53 (and other molecular markers) as a research factor and as a new diagnostic test for our cancer patients. Essentially, we know from cited studies that P53 controls the apoptotic pathway and can play a key role in cancer cell self-destruction. We also know from the growing body of evidence that mutated or defective P53 can alter the efficacy of antineoplasic agents.2
Initially, my idea was first to check out the connection between cancer and P53 mutation as described in the literature and what could be done about it. Of course, it was necessary to teach patients about the meaning of the P53 apoptotic pathway and its relation to cancer. We prepared leaflets about the P53 tumor suppressor gene and testing procedure to educate our patients. In the beginning, patients were reluctant to have this test; after fully understanding the benefit of P53 testing, most were in favor of it.
After 2 years observing P53 gene expression and P53 protein level tests from about 400 patients with different types and grades of cancer, we actually found that more than half exhibited the P53 mutation. For breast cancer cases, 50% tested for mutation (not 25% as described in the literature), while others exhibited a defective P53 function.
While the P53 gene is active, it only produces low level P53 protein, which is not sufficiently high to self-destroy abnormal cells. An inactivation of normal (wild type) P53 genes with very low protein production may offer the same unfavorable result as would a mutation, since transformed cells are not fully self-destroyed, accumulate further mutations, favoring the transformation toward tumor growth. Not all cancers harbor a P53 mutation, but low protein production contributes to cancer development and resistance to tumor chemotherapy drugs, even with the newest agents.3
At this point, I want to make clear the apoptotic effects of certain dietary agents such as curcumin and resveratrol. When we say that they induce apoptosis, it only occurs in cells expressing wild-type P53, not in P53 mutated cells. This is not always clearly explained and can be misleading when it comes to treatment.
We also learned from many studies that by targeting P53 and reverse mutation, we have a therapeutic application, which can be seen as an anticancer therapy, increasing the effectiveness of chemotherapy.4,5 We know that chemotherapy doesn't directly kill cancer cells but rather destroys them by inducing apoptosis through the cell's cycle arrest and the activation of the P53 cellular death channel.
Cancer cells with mutated P53 are resistant to most forms of treatment that target self-destruction by apoptosis, including chemotherapy unless the P53 pathway is reactivated. P53 mutation in malignant breast cancer increases the aggressiveness of the disease and risk of death.6 Unfortunately, few patients were unable to understand the meaning of their tests with mutated P53 and preferred to pursue chemotherapy, and even those choosing conventional therapy died because of poor response and damaging effects of toxic agents. Again, the P53 test can only check for the efficacy of chemotherapeutic agents and predict reactive P53 gene expression before the patient starts treatment. I have a case wherein a 36-year-old well-known musician was under family pressure to seek medical treatment. She opted solely for chemotherapy, despite that her P53 test that showed mutation and poor prognosis. About 6 months later, I heard on the evening news about her death – which was tragic, since I had warned the family about the consequences if we didn't treat her first in order to reverse her P53 mutation.
Genetic Correction Strategies
Our second objective was to see if P53 mutation could be reversed to restore oncogenic function, activating self-destruction of cancer cells. Obviously, for the first time we demonstrated that selected natural compounds made from small molecules, nucleic acids, and antioxidants can restore P53 function by targeting mutant P53 with significant therapeutic application. Indeed, we developed a "genetic corrective strategy," a breakthrough in cancer treatment.
This new strategy selectively increases self-destruction of mutant cancer cells but not healthy cells with a normal P53 function. With our cancer therapy, we improve both survival time and quality of life of cancer patients. We determined that this treatment either reduces or eliminates the risk of disease recurrence.
After having fully experimented with P53 testing and P53-based cancer therapy, we intend to investigate other molecular markers that correlate with cancer development as follows:
- P53 gene expression
- BAX gene expression
- BCL2 gene expression
- Survivin gene expression
- P21 gene expression
We will demonstrate that not only P53 but other molecular markers provide full information about cancer development, and offer both evaluation and predictive value for breast cancer patients.7,8
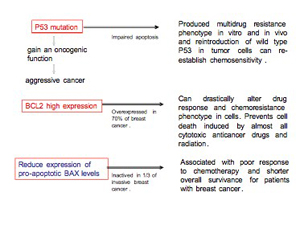
Molecular Markers
Evidence has shown that the proapoptotic gene BAX that mediates apoptosis through the mitochondria becomes inactive in one-third of invasive breast cancers. In a study of 119 women with metastatic breast cancer, patients whose tumors had lost BAX activity also had a poor response to combination chemotherapy, a faster time line to tumor progression, and shorter overall survival, whereas enhanced expression of BAX protein correlates with a good response to chemotherapy in vivo.9,10 There is a relationship between P53 and BAX in signaling apoptosis, but some of the failure of P53 to stimulate BAX protein is due to antiapoptotic protein BCL2 activity, which acts to inhibit apoptotic protein which upregulates in 70% of breast cancers and correlates with resistance of cancer cells to a wide spectrum of chemotherapeutic agents.11,12 Overexpression of the antiapoptotic BCL2 protein blocks cytochrome C release in response to a variety of apoptotic stimuli, whereas the proapoptotic BAX protein stimulates cytochrome C to activate an apoptotic cascade.
We ourselves have observed that this multiresistance to drugs involves P53 mutation, low BAX activity, and highly activated BCL2. Not only is this common in primary breast cancer but is usually exhibited in cancer recurrence, along with the risk of metastasis. Therefore, not only P53 but also BCL2 may both be seen as offering prognostic value in breast cancer, becoming an attractive target in developing new therapeutic approaches.13,14 Over the years, we have been successful in targeting as well as decreasing highly expressed BCL2 or inhibiting its activity to eliminate cancer cells' resistance, activating their self-destruction.
Survivin is another highly expressed gene in most cancers, but not in healthy tissue. It plays a key role in the development and aggressivity of breast cancer.15,16 Survivin is virtually absent from normal breast tissues, but mostly present at very high levels in malignant breast tumors.17 Survivin inhibits apoptosis with the apoptotic enzymes, and caspase-3 and -7 activation, leading to negative regulation of apoptosis and cancer cells' resistance.18,19 Increased expression of survivin is associated with chemotherapy resistance, increased rate of tumor recurrence, and shorter patient survival.20
In a study of over 500 breast cancer patients conducted by Brid Ryan at the University College Dublin Department of Biochemistry, it was found that patients with high levels of survivin had reduced survival times and were more likely to have tumor recurrence; thus a strong predictor of poor prognosis in breast cancer patients.21 Increased survivin expression is associated with angiogenesis, since survivin expression protected endothelial cells from apoptosis during the proliferative and remodeling phases of angiogenesis. Some new studies have shown that targeting survivin to breast cancer brain metastases may well be an effective antiangiogenic therapy, since many drugs cannot enter the brain because of its impermeable blood vessels.22
This serves as one other reason for tumors' resistance characterized by strong neovascularization, making more difficult the task of killing these cells with chemotherapeutic drugs. Also, it is suggested that survivin may be involved in driving the growth of tumor cells by sending a signal that allows them to develop at a very high rate.
Overall survivin may serve as a prognostic marker for breast cancer and recurrence risk as well as for attractive new cancer interventions.23 As described through our observation and molecular marker tests, survivin is overexpressed in 60% of primary breast cancer with metastases, and I would propose a similar percentage concerning disease recurrence.
Monitoring P53, BAX, and survivin gene expression in primary breast tumors may be a relevant prognostic marker for tumors' resistance and may indicate as well the strong angiogenic activity and microvessel density.24 Therefore, a strategy is required to inhibit and reduce angiogenesis activity, which in turn may reduce tumor growth and prevent metastatic invasion.
We have observed that in breast cancer, highly expressed survivin may be a very bad prognostic for patients with shorter survival time, especially with poor response to chemotherapy and faster metastatic invasion. However, we have been successful in targeting expressed survivin and therefore have increased the rate of remission or prevention from imminent breast cancer recurrence.
P21 is a gene that promotes the self-destruction of cells damaged by toxic agents, including radiation. Elevated levels of P21 indicate that cells are being exposed to an attack by toxic agents and will self-destruct, which is positive in the presence of cancer and radiation treatment. Low levels of P21 indicate that the self-destruction process is not operating properly and many harmful cells survive, representing a poor prognosis. P21 is associated with the second line of cellular defense (after the P53 gene) against the growth of the cancer cell population. Lack of P21 expression has been related to poor prognosis in several solid tumor types with metastases.25
P21 and survivin gene expression work together, and when P21 is highly active, survivin is usually less activated and vice versa. For instance, if the survivin gene expression is normal and P21 gene expression is high, a radiotherapy regimen will be effective. However, if survivin gene expression is low and P21 gene expression is low, the radiotherapy regimen will not be effective. If survivin gene expression is high and P21 gene expression is low, the radiotherapy regimen will not be effective. BAX and BCL2 represent one type of cancer cells, while survivin and P21 represent another type, and evaluation between the two respective genes is important to establish as diagnostic and prognostic.
Tumor Marker 2:
Pyruvate Kinase (TM2-PK)
Another interesting novel molecular marker is TM2-PK, a key regulator of the metabolic alteration found in tumor cells. TM2-PK is the glycolytic isoenzyme that is overexpressed in a wide range of tumor cells. This enzyme plays a significant role in the metabolic process in which cancer cells generate needed energy. As such, TM2-PK is a strong marker for the presence of metabolically active tumor growth.26,27 Elevated levels are associated with the presence of a primary tumor, while a level slightly above reference range indicates a situation potentially at risk – very low levels reflect cancer cells' use of the body's fat or protein store as an energy source, a process that can severely damage general metabolism.
We usually perform the TM2-PK level test on our cancer patients at the first consultation and on patients whom we believe may have a risk of cancer. You will be surprised by the result. We then perform a second test after 30 to 60 days after P53 has been activated to check if cancer cells are less active. There is also a way to determine the quantity of dying cancer cells from the applied treatment by having the patient take the extracellular telomerase and intracellular telomerase test. We know that telomerase activity is overexpressed in most cancer cells so that they may become immortal and resistant to most forms of destruction.28 We know that an activated P53 pathway led to the appearance in the blood of dying cancerous cells that then are detected by measuring the telomerase activity of patients. It may also indicate evidence of the tumor's disintegration.
By performing the extracellular telomerase and intracellular telomerase testing, after activation of P53 expression and decreased antiapoptotic gene activity, we have been able to prove that our treatment – without conventional therapy – can target and destroy cancer cells, which is a new advance in treating cancer with nontoxic treatment. It also shows how important it is to evaluate molecular markers, since it may serve in diagnostic, prognostic, and predictive value.29 (For more information and reference range of the molecular markers see www.sergejurasunas.com, "Diagnostics.")
Figure 1

Figure 2
Advantage of the Genetic Blood Assay
This package of a complete molecular markers permits the physician to know the full scenario from the outset and therefore introduce a better treatment strategy, wherein from time to time s/he can check from a scientific standpoint whether the treatment is effective.
1. Do we have a declared cancer?
2. Do we have one or more population of resistant cancer cells?
3. Do we have resistant cancer cells?
4. Do we have a protumoral activity over a antitumoral activity?
5. Do we have a antitumoral activity?
6. Do we have a positive effect from treatment protocols?
7. What is the percentage of cancer cell's self-destruction or tumor disintegration from the treatment?
Restoration of P53 Function: Targeting Mutant P53 and Other Molecular Markers
One of the new therapeutic strategies in cancer is the option to restore P53 functionality and to activate the production of P53 proteins, which in turn increase self-destruction of cancer cells and may even lead to tumor regression.30 Other attractive targets include BAX, BCL2, survivin, and P21 manipulation. To illustrate this article we would like to offer some interesting results based upon several years of our clinical work, encompassing molecular marker testing on breast cancer patients, disease recurrence or imminent recurrence, followed by targeting molecular targeting therapy.31
Examples:
Red Color: Mutated protein (harmful)
Black color: Normal protein
Figure 3
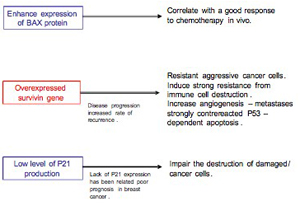
Figure 4
Breast cancer recurrence with distant metastases to lung and liver
Figure 5

Comments
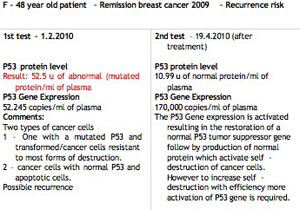
The P53 tumor suppressor gene and P53 protein level are totally inactive and show that apoptosis is not functioning. A proliferation of resistant cancer cells can be expected, while survivin gene expression is very high. The ratio of survivin/P21 genes indicates that a pool of resistant cancer cells may accumulate in the blood. The only antitumor cellular activity is mainly dependent on the P21 activity, which is not enough to arrest the growth of the cancer cell population.
The TM2-PK result indicates a metabolically active tumor but not very high at the moment. However, the context is not favorable for chemotherapy effectiveness and the patient followed several lines of chemotherapy regimen including 5 Fu, Cytofosfamide, Pacitaxel, Herceptin, etc., without success.
This is a poor prognosis.
Figure 6

Figure 7

Female, 63 years old; breast cancer remission with high risk of recurrence treated to target highly expressed antiapoptotic proteins and actived P53 pathway.
Figure 8

Figure 9
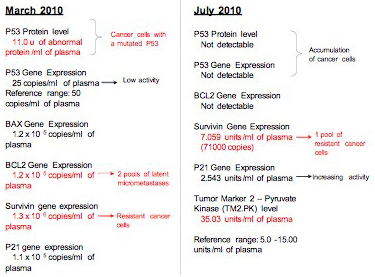
Figure 10

Conclusions
Today we have opened a new way to contribute enormously to anticipating and preventing cancer by monitoring patients with P53 testing and other related molecular markers. P53 has a central role in the control of cell growth and apoptosis and frequent mutations in a tumor, making it a unique target for cancer therapy. In addition, P53 activity in normal cells may protect them from the side effects of chemotherapy or radiation. Over the years, we have worked with P53 and other molecular markers directly with cancer patients and observed how they may or may not improve in function of the molecular marker tests, as seen with several examples included in this article. What is important is that breast cancer can be prevented, while breast disease recurrence can be prevented as well. This helps overall to avoid more trauma and often a poor outcome.
In addition, several new natural compounds and combination therapies, including low molecular antioxidants, nucleic acids, and peptides, have demonstrated efficacy in targeting P53 and other related molecular markers, which is a breakthrough in naturopathic oncology.
We now have a greater possibility to select the best treatment protocols with positive effects, and follow the decreasing tumor's activity and the percentage of cancer cell self-destruction. This means that for the first time you can prove the efficacy of your treatment, and observe improvement step by step.
By administering P53 testing together with BAX, BCL2, survivin, and P21 gene expression tests, we are in a position to know which patient may improve quickly with no metastatic condition and, conversely, which patients already have metastasic risk or dissemination. If you consult my website's section on Diagnostics (http://www.sergejurasunas.com/index.php?
option=com_content&view=article&id=12&Itemid=13), one example is the case of a patient in whom the result from molecular testing had shown only a relatively small proportion of cancer cells (about 25%) under self-destruction. By inhibition of the antiapoptotic protein and activation of proapoptotic protein and overall apoptotic pathway, we can scientifically control the disease and the treatment prescribed to patients.
Of course, this requires choosing the adapted treatment that first is always experimental, and this is what I did. Personal experience is always necessary if we want to discover the best treatment for our patients, even if we are not successful at the beginning. Sometimes you may have difficulty in reactivating a mutated P53 expression even by using the selected compounds because you may find out that the patient has an emotional disorder involving neurotransmitters and an exhausted immune system, leading to a bacterial infection responsible for the mutation. But this is the beauty of the healing art that we are practicing, which requires experience and intuition to achieve the best results.
Information
The reader may be confused with the P53 gene expression results, first in understanding the term copies and then the term units, which totally depends on the testing laboratory. 1 unit is approximately 1.000 copies. As mentioned before, the reader may wish to obtain more information about molecular markers and reference range as well as information about breast cancer and the P53 tumor suppressor gene. Consult www.sergejurasunas.com.
The original article with more complete information and illustrations could not be published here due to space limitations. It is available for Townsend Letter readers at www.sergejurasunas.com.

Notes
1. Lee Y-TN. Breast carcinoma. Pattern of recurrence and metastasis after mastectomy. Am J Clin Oncol. 1984;7:443–449.
2. Bunz F, Hwang PM, Torrance C, et al. Disruption of the P53 in human cancer cells alters the responses to therapeutic agents. J Clin Invest. 1999;104:263–269.
3. Gottesman MM, Fojo T, Bates SE. Multidrug resistance in cancer: Role of ATP-dependent transporters. Nat Ver Cancer. 2002;2:48–58.
4. Stokasa T, Golab J. Prospect for P53-based cancer therapy. Acta Biochim. 2005;52(2):321–328.
5. Selivanova G, Kawasaki T, Ryabchenko L, Wiman KG. Reactivation of mutant P53: A new strategy for cancer therapy. Semin Cancer Biol. 1998;8:369–378.
6. de Moura-Gallo CV, Simão TA, Ribeiro FS, Andrade-Serpa MJ, Cardoso LEB, Mendonça GAS. TP53 mutation in malignant breast tumors: association with risk factors and clinical-pathological characteristics, including risk of death, in patients from Rio de Janeiro (Brazil). Int J Cancer. 2002;101:69–73.
7. Son M-W, Woo H-D, Sohn D-M, et al. Prognostic value of apoptosis and the survivin, BCL2 and P53 expression in breast cancer patients. J Breast Cancer. March 2007;10(1):19–28.
8. Malamou-Mitsi V, Gogas H, Dafni U, et al. Evaluation of the prognostic and predictive value of P53 and BCL2 in breast cancer patients participating in a ramdomized study with dose-dense sequential adjuvant chemotherapy. Ann Oncol. 2006;17(10):1504–1511.
9. Isrid Sturn et al. Impaired BAX protein expression in breast cancer: Mutational analysis of the BAX and the P53 gene. Int J Cancer. 24 July 2000.
10. Zhang X-K. TR3-based peptides for apoptosis in breast cancer. California Breast Cancer Research program. Initial Award Abstract; 2002.
11. Chipuk JE, Kuwana T, Bouchier-Hayes, et al. Direct activation of BAX by P53 mediates mitochondrial membrane permeabilization and apoptosis. Science. 13 February 2004;303(506):1010–1014.
12. Zhang 2002.
13. Malamou-Mitsi et al. 2006.
14. Marza, Naval J. BCL2 family members as molecular targets in cancer therapy. Biochem Pharmacol. 15 October 2008;76(8):939–946.
15. Ambrosini G, Adida C, Alterieri DC. A novel anti-apoptotic gene, Survivin, expressed in cancer and lymphoma. Nature Med. 1997;3:917–921.
16. Kennedy SM, O'Driscoll L, Purcell R, et al. Prognostic importance of Survivin in breast cancer. Br J Cancer. 2003;88:1077–1085.
17. Ryan B. Surviving breast cancer. Irish Sci Yearb. 2004.
18. Shin S, Sung BJ, Cho YS, et al. An Anti-apoptotic protein human Survivin is a direct inhibitor of Caspase 3 and 7. Biochemistry. 2001;40(4):1117–1123.
19. Tamm J, Wang Y, Sansville E, et al. IAP-family protein Survivin inhibits caspase activity and apoptosis induced by Fas (CD95), Bax, caspases, and anticancer drugs. Cancer Res. 1998;58(23):5315–5320.
20. Kawasaki H, Altieri DC, Lu CD, Toyoda M, Tenjo T, Tanigawa N. Inhibition of apoptosis by Survivin predicts shorter survival rates in colorectal cancer. Cancer Res. 1998;58:5071–5074.
21. Ryan 2004.
22. Hofman F. Survivin: target for breast cancer brain metastases. University of Southern California. Initial Award Abstract 2005 (online).
23. Zaffaroni N, Pennati M, Dardone MG. Survivin as a target for new cancer interventions. J Cel Mol Med. 2005;9(2):360–372.
24. Gasparini G, Weidner N, Bevilacqua P, et al. Tumor microvessel density, P53 expression, tumor size and peritumoral lymphatic vessel invasion are revelant prognostic markers in node-negative breast carcinoma. J Clin Oncol. 1994;12:454–466.
25. Ito K, Sasano H, Matsunaga G, et al. Correlation between P21 expression and clinicopathological finding, P53 gene and protein alterations, and survival in patients with endometrial carcinoma. J Pathol. 1997;183:318–324.
26. Kaura B, Bagga R, Patel FD. Evaluation of the Pyruvate Kinase Isoenzyme tumor (Tu M2-PK) as a tumor marker for cervical carcinoma. J Olistet Gynaecol Res. 2004;30:193–196.
27. Luftner D, Maesterhaw J, Akwivakis C, Geppert R, Petrides PE, Possinger K. TM2-PK expression in advanced breast cancer. Anticancer Res. 2000;20:5077–5082.
28. Kim NW, Piatyszek MA, Prowse KR, et al. Specific association of human telomerase activity with immortal cells and cancer. Science. 1994;266:2011–2015.
29. Roas PH. Molecular markers as diagnostic, prognostic and susceptibility predictors in bladder cancer. Eur Urol Rev. 2008:34–38.
30. Ventura A, Kirsch DG, McLaughlin ME, et al. Restoration of P53 function leads to tumor regression in vivo. Nature. 2007;445:661–665.
31. Jurasunas S, Taylor OG. How to target mutant P53 in a case of multiple cancer recurrence. Townsend Lett. August/Sept. 2010;325:26.

 Serge Jurasunas is an internationally well known doctor of naturopathy and alternative medicine with over 40 years of experience in the treatment of cancer. He is developing innovative therapies in cancer treatment and is a pioneer in live blood analysis, dried blood oxidative stress, and iridology. Serge Jurasunas is an internationally well known doctor of naturopathy and alternative medicine with over 40 years of experience in the treatment of cancer. He is developing innovative therapies in cancer treatment and is a pioneer in live blood analysis, dried blood oxidative stress, and iridology.
Dr. Jurasunas is busy working with P53 tumor suppressor gene and other molecular markers testing related to cancer patients and patients with high risk of cancer.
For more information and to learn about cancer treatment, molecular markers, and clinical cases at Holiterapias Clinic, please visit www.sergejurasunas.com; e-mail: info@sergejurasunas.com; phone: +351 213471117.

|



![]()
![]()












 Serge Jurasunas is an internationally well known doctor of naturopathy and alternative medicine with over 40 years of experience in the treatment of cancer. He is developing innovative therapies in cancer treatment and is a pioneer in live blood analysis, dried blood oxidative stress, and iridology.
Serge Jurasunas is an internationally well known doctor of naturopathy and alternative medicine with over 40 years of experience in the treatment of cancer. He is developing innovative therapies in cancer treatment and is a pioneer in live blood analysis, dried blood oxidative stress, and iridology.